How Intermolecular Forces Affect Phases of Matter
Key Questions
-
As mentioned here, intermolecular forces (IMFs) are important because they are the leading cause for differences in physical properties between similar molecules.
Be sure to read the linked answer to review if you are unfamiliar with IMFs.
Physical properties commonly discussed when relating to IMFs in pure substances are:
- Melting and boiling points - when molecules go from solid to liquid or liquid to gas.
- Vapor pressure - the pressure exerted by gases onto the walls of the container
- Enthalpy of vaporization - energy needed at constant pressure to turn a liquid to gas
- Viscosity - thickness of a liquid when it comes to fluid flow
- Surface tension - resistance to distortion at the surface of the liquid from poking
The core principle is that the stronger the IMFs in the sample of molecules, the more strongly they interact, which means they stick together more.
That leads to the following trends:
- Stronger IMF
->→ Higher melting and boiling points (harder to melt and boil) - Stronger IMF
->→ Lower vapor pressure (harder to boil) - Stronger IMF
->→ Higher enthalpy of vaporizationDeltaH_(vap) (takes more energy at constant atmospheric pressure to turn liquid to gas) - Stronger IMF
-> Higher viscosity (a thicker liquid, flows more like molasses) - Stronger IMF
-> Higher surface tension (more resistant to deformation from poking)
-
Intermolecular forces (IMFs) are attractive interactions between molecules. They lead to differences and sometimes trends in various physical properties.
These are typically listed in order of strength:
"Dispersion" < "Dipole-Dipole" < "Hydrogen-bonding" < "Ion-Dipole" < "Ion Pairing" Hydrogen-bonding (which is not bonding) is shown below in an example diagram for
"H"_2"O" and"HF" .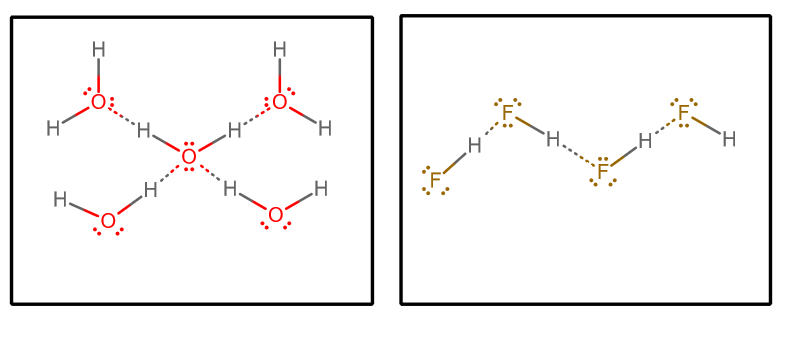
Other features which are equally important are described below.
IMFs span a spectrum of:
- temporary partial charge-partial charge interactions
- permanent partial charge-partial charge interactions
- permanent partial charge-full charge interactions
- permanent full charge-full charge interactions
Depending on electronegativities of each atom involved, the IMF strength and interaction time changes.
Here are tables organizing this into the known IMFs.
Table 1. Interaction Times
ul("IMF"" "" "" "" "" "" "" ""Interaction Time")
" "" "" ""Dispersion"" "color(white)(....)"Temporary"
" "" ""Dipole-Dipole"" "" ""Permanent"
"Hydrogen-bonding"" "color(white)(....)"Permanent"
" "" "" ""Ion-Dipole"" "color(white)(....)"Permanent"
" "" "" ""Ion Pairing"" "" ""Permanent" Table 2. Degree of Charge Interactions
ul("IMF"" "" "" "" "" "" "" ""Degree of Interaction")
" "" "" ""Dispersion"" "color(white)(....)"Partial/Partial"
" "" ""Dipole-Dipole"" "" ""Partial/Partial"
"Hydrogen-bonding"" "color(white)(....)"Partial/Partial"
" "" "" ""Ion-Dipole"" "color(white)(....)"Full/Partial"
" "" "" ""Ion Pairing"" "" ""Full/Full" Table 3. Typical Participants
ul("IMF"" "" "" "" "" "" "" ""Participants"" "" "" "" "" "" ")
" "" "" ""Dispersion"" "" ""Nonpolar Molecules"
" "" ""Dipole-Dipole"" "color(white)(..)"Moderately Polar Molecules"
"Hydrogen-bonding"" "color(white)(..)"Molecules with XH bonds"^"*"
" "" "" ""Ion-Dipole"" "" ""Ions in Polar Solvent"
" "" "" ""Ion Pairing"" "color(white)(..)"Ions with large charges" "*" - "X" indicates a very electronegative atom, usually"N" ,"O" , or"F" . Note that these must be bonded DIRECTLY to"H" , not just be in the chemical formula. Note also that there are exceptions, like chloroform interacting with acetone, which do NOT seem to contain sufficiently electronegative atoms.