What are some examples of theoretical chemistry?
1 Answer
Theoretical chemistry tends to involve events that can't be easily observed in real life, such as radical reactions, short-lived reaction intermediates, gas-phase reaction dynamics, etc. It can also involve more realistic events that are calculated to save money for the experimentalists.
This can get pretty out-there...
Here are some example published papers where I've distilled the information down into basic...ish summaries.
Theoretical Advanced Inorganic Chemistry
- Can Multiconfigurational Self-Consistent Field Theory and Density Functional Theory Correctly Predict the Ground State of Metal–Metal-Bonded Complexes? by Laura Gagliardi et al.; University of Minnesota
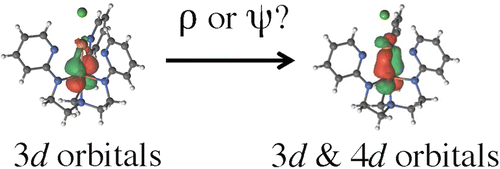
This paper uses ab initio computational approximations (basically, generating approximate models of the orbital configurations using first principles) to investigate the electronic structure of transition metal complexes in which a metal-metal bond exists.
These might include wave-function-based (
#psi# ) methods like multiconfigurational second-order perturbation theory restricted active space (RASPT2) and density-based (#rho# ) methods such as density functional theory (DFT). The former is indicated as#psi# and the latter is indicated as#rho# in the diagram.These investigated compounds include for instance, those that have
#"Fe"-"Fe"# bonds,#"Fe"-"Mn"# bonds, etc., wherein the#d# orbitals are used to bond directly.
Theoretical f-block Chemistry
- A combined photoelectron spectroscopy and relativistic ab initio studies of the electronic structures of
#"UFO"# and#"UFO"^(-)# by Kirk Peterson et al.; Washington State University
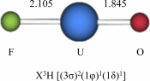
This paper uses high-level ab initio computational chemistry methods in conjunction with experimental photoelectron spectroscopic studies to investigate the electron correlation within two triatomic uranium-based compounds.
(Plus, it's a great excuse to publish a paper with the word "UFO" in it!).
This dynamical and nondynamical correlation, due to the closeness of adjacent energy levels, includes either short-range electron repulsions, or multiple degenerate electron configurations in the
#5f# and (maybe#6d# ) orbitals.(These near-degeneracies basically make it take a lot longer to model these systems accurately.)
Theoretical Radical Chemistry
- Design of Leaving Groups in Radical
#"C"-"C"# Fragmentations: Through-Bond#2c-3e# Interactions in Self-Terminating Radical Cascades by Igor Alabugin et al.; Florida State University
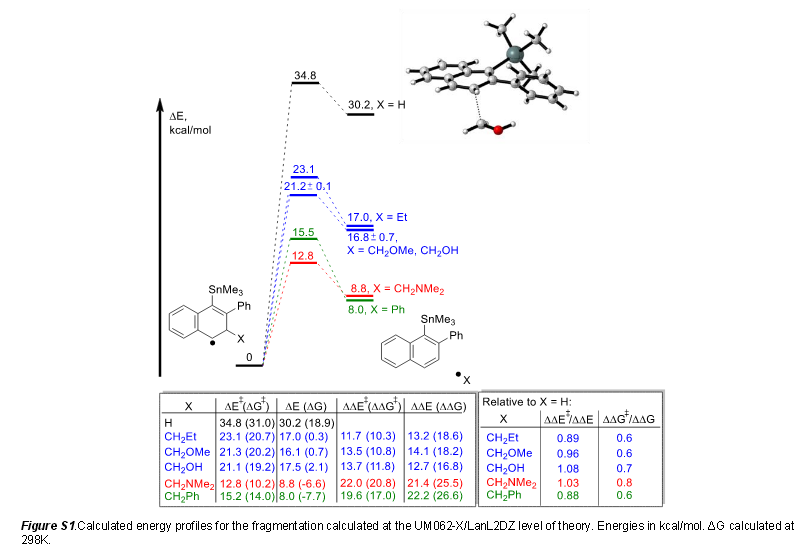
Basically, computational modeling was used to predict which leaving groups are best for particular aromatic radical reactions involving
#"Sn"# , such that the transition state is stabilized and the reaction is capably terminated via a#beta# -scission (elimination of a radical fragment to form a double bond).Based on my experience having met Dr. Alabugin in person, I believe this was in conjunction with actual experiment, so that chemical resources are conserved. :)
(That's a common theme of theoretical chemistry: to save money for the experimentalists.)

