What are the four quantum numbers?
1 Answer
See below.
Explanation:
The four quantum numbers are the principle quantum number,
The principle quantum number ,
For example, the
#3d# subshell is in the#n=3# shell, the#2s# subshell is in the#n = 2# shell, etc.
The angular momentum quantum number ,
For example, the
#n=3# shell has subshells of#l=0,1,2# , which means the#n=3# shell contains#s# ,#p# , and#d# subshells (each containing their respective orbitals). The#n=2# shell has#l=0,1# , so it contains only#s# and#p# subshells. It is worth noting that each shell has up to#n-1# types of subshells/orbitals.
The magnetic quantum number ,
So, for a
#2p# orbital with#n=2# and#l=1# , we can have#m_1=-1,0,1# . This tells us that the#p# orbital has#3# possible orientations in space.If you've learned anything about group theory and symmetry in chemistry, for example, you might remember having to deal with various orientations of orbitals. For the
#p# orbitals, those are#p_(x)# ,#p_(y)# , and#p_(z)# . So, we would say that the#2p# subshell contains three#2p# orbitals (shown below).
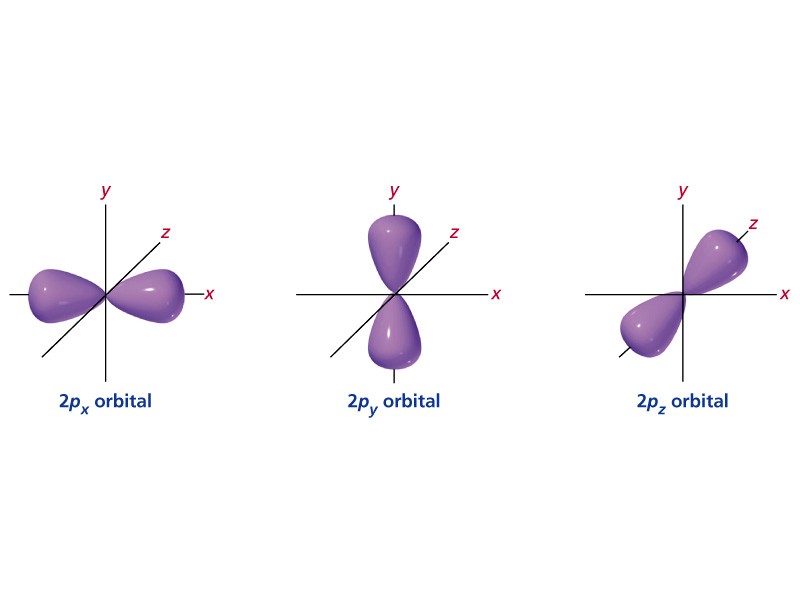
Finally, the electron spin quantum number,
Remember that there are only two electrons to every orbital, and that they should have opposite spins (think Pauli exclusion principle). This tells us that there are two electrons per orbital, or per
#m_l# value, one with an#m_s# value of#+1/2# and one with an#m_s# value of#-1/2# .
Therefore,

