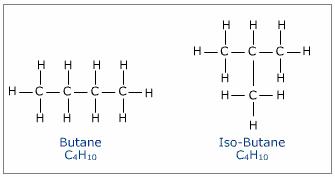
What are the isomers of butane?
1 Answer
Feb 15, 2015
Butane, or
As you know, isomers are molecules that have the same molecular formula but different chemical structures. In the case of butane, its two isomers will have these structural formulas
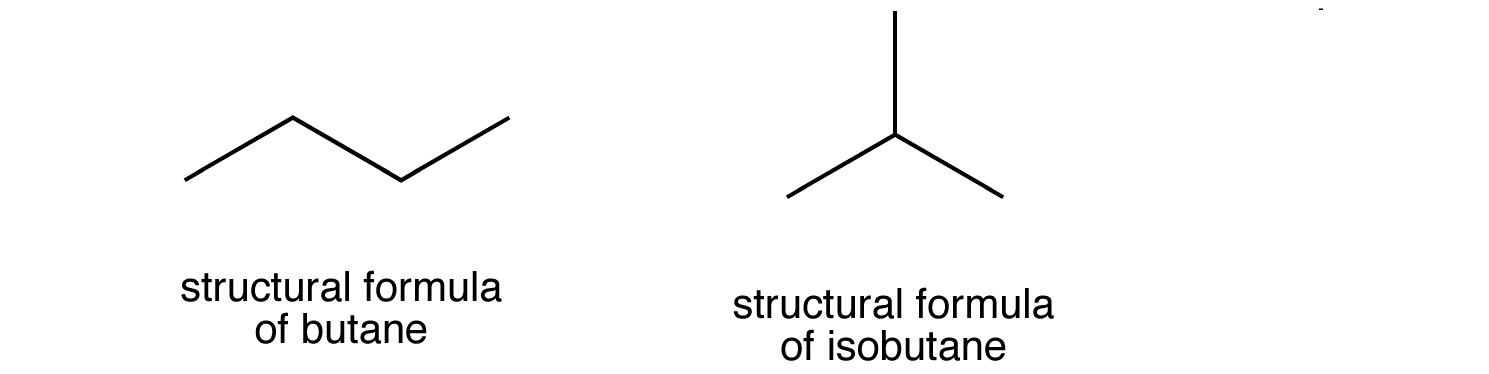
Notice that isobutane has a propane parent chain with a methyl group -