Isomerisms
Key Questions
-
Short answer: Isomers have little to do with understanding the diversity of life.
Isomers are compounds that have the same chemical formula but different structural formulas.
One of the reasons for biodiversity is genetic variations.
Genes are subunits of DNA. Each gene contains a particular set of instructions. It usually codes for a particular protein.
There are only four different chemical bases in a gene (adenine, thymine, cytosine, and guanine). The order of the bases determines the function of the gene.
Each gene consists of thousands, even hundreds of thousands, of chemical bases.
If two of these genes had exactly the same number of each type of base, then they would be isomers of each other.
But that is extremely unlikely. If a gene has just one different base or one more base, it is no longer an isomer. It is a different compound.
Isomerism has little to do with the diversity of life.
-
Before I define what isomers are ,I will give you a simple example, think that you have three circles,each of same color , same radius and same mass. You can arrange the three circles by putting one next to another, or you can arrange the three one overlapping the other. In both the arrangements has same mass , same color but what differs is the arrangement of circles. This defines isomers.
Isomers are molecules with the same chemical formula but different chemical structures. That is, isomers contain the same number of atoms of each element, but have different arrangements of their atoms in space.
A simple example of isomerism is given by propanol: It has the formula
#C_3# #H_7# OH and occurs as two isomers: propan-1-ol (n-propyl alcohol; I) and propan-2-ol (isopropyl alcohol; II)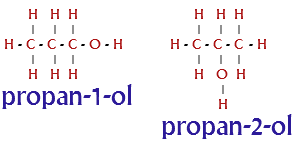
the two isomers have same molecular formula, same mass , but differs in the arrangement of Hydroxyl (OH ) group.