What are the names of the isomers for C4H8Br2?
1 Answer
Nov 24, 2015
Well, I can find 9 isomers, if we do NOT count R/S.
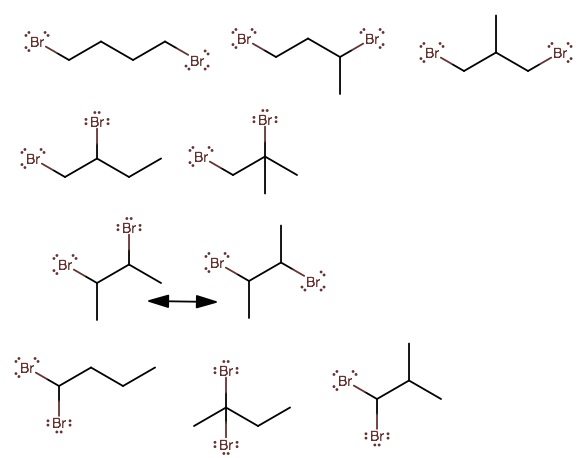
The 2,3-dibromobutane would have free bond rotation about the C2-C3 bond, which is indicated with the "resonance" double arrow.
The general process was to shift the bromide around while taking symmetry into account, and then shift the methyl around while accounting for symmetry, and fill in any gaps you have left.
Names:
1,4-dibromobutane
1,3-dibromobutane (+ R/S)
1,3-dibromo-2-methylpropane
1,2-dibromobutane (+ R/S)
1,2-dibromo-2-methylpropane
2,3-dibromobutane (+ R/S)
1,1-dibromobutane
2,2-dibromobutane
1,1-dibromo-2-methylpropane

