What are the oxidation numbers of 2Cl- and Cl2?
1 Answer
for the
for the
Explanation:
The chemical symbol
The overall oxidation state of the 2 moles
Now for
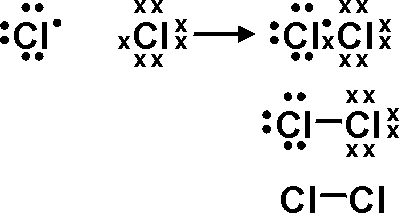
The sharing of electrons brings a formal charge of +1 for one of the
for the
for the
The chemical symbol
The overall oxidation state of the 2 moles
Now for
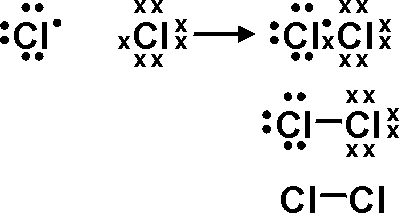
The sharing of electrons brings a formal charge of +1 for one of the