What are the phase diagrams of water and carbon dioxide?
1 Answer
Nov 23, 2017
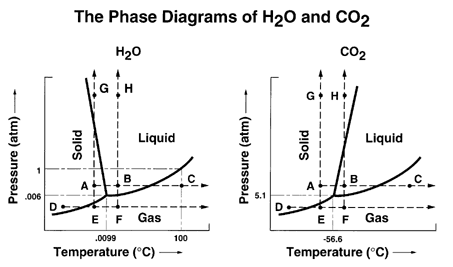
Each line represents phase changes between respective boundaries. The triple point is where all the phases are at equilibrium.
Phase changes are usually isothermal, so temperature is constant until every molecule has attained high enough heat (e.g. kinetic energy) to surpass the intermolecular forces characteristic of each phase at varying strengths.

