What are the products in the following reaction?
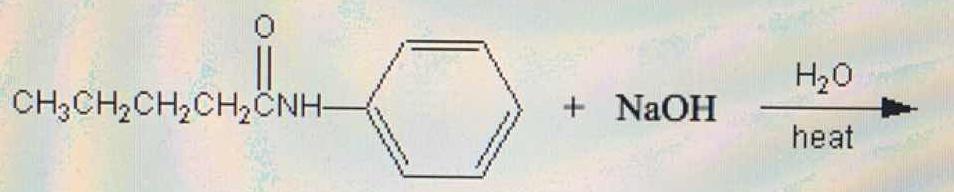
1 Answer
Mar 12, 2017
Explanation:
So the prodcuts are water, the amine, and carboxylate salt.
Note that to make the amide, typically we would take the acid chloride, AND TWO equivs of the amine (or if the amine were expensive we would use 1 equiv of amine and 1 equiv of

