What are the products of this reaction: #Zn(s) + Pb(NO_3)_2(aq) -> ??#
1 Answer
The products are the element lead, and the compound zinc nitrate,
Explanation:
This is a single replacement (also called a single displacement) reaction involving the metals zinc and lead.
The generic form for a single replacement reaction involving metals, is:
However, not all reactions represented as single replacements will occur. The metal that replaces the other metal in the compound must be more reactive than the one that is replaced.
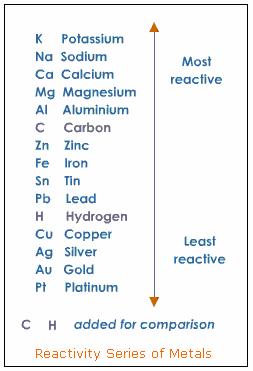
We can determine whether a single replacement will occur by checking a Metal Activity Series like the one below. Any metal can replace a metal beneath it, but not above it.
As you can see, zinc is above lead in the series, which means that zinc can replace all of the metals below it in the series, including lead. So the single replacement reaction will occur.