What does an NMR spectrum tell you?
1 Answer
NMR spectra tell us how many
Explanation:
Modern NMR spectrometers can do many weird and wonderful things.
I shall limit this discussion to One-Dimensional NMR Spectroscopy, in which frequency is plotted along the horizontal axis and signal intensity is plotted along the vertical axis.
- Chemical shift gives type of
#"H"# atom. - Integration gives numbers of each type of
#"H"# atom - Splitting tells which
#"H"# atoms (and how many) are on adjacent C atoms.
(a) Broad Band Decoupled Spectra
These are the "normal" spectra.
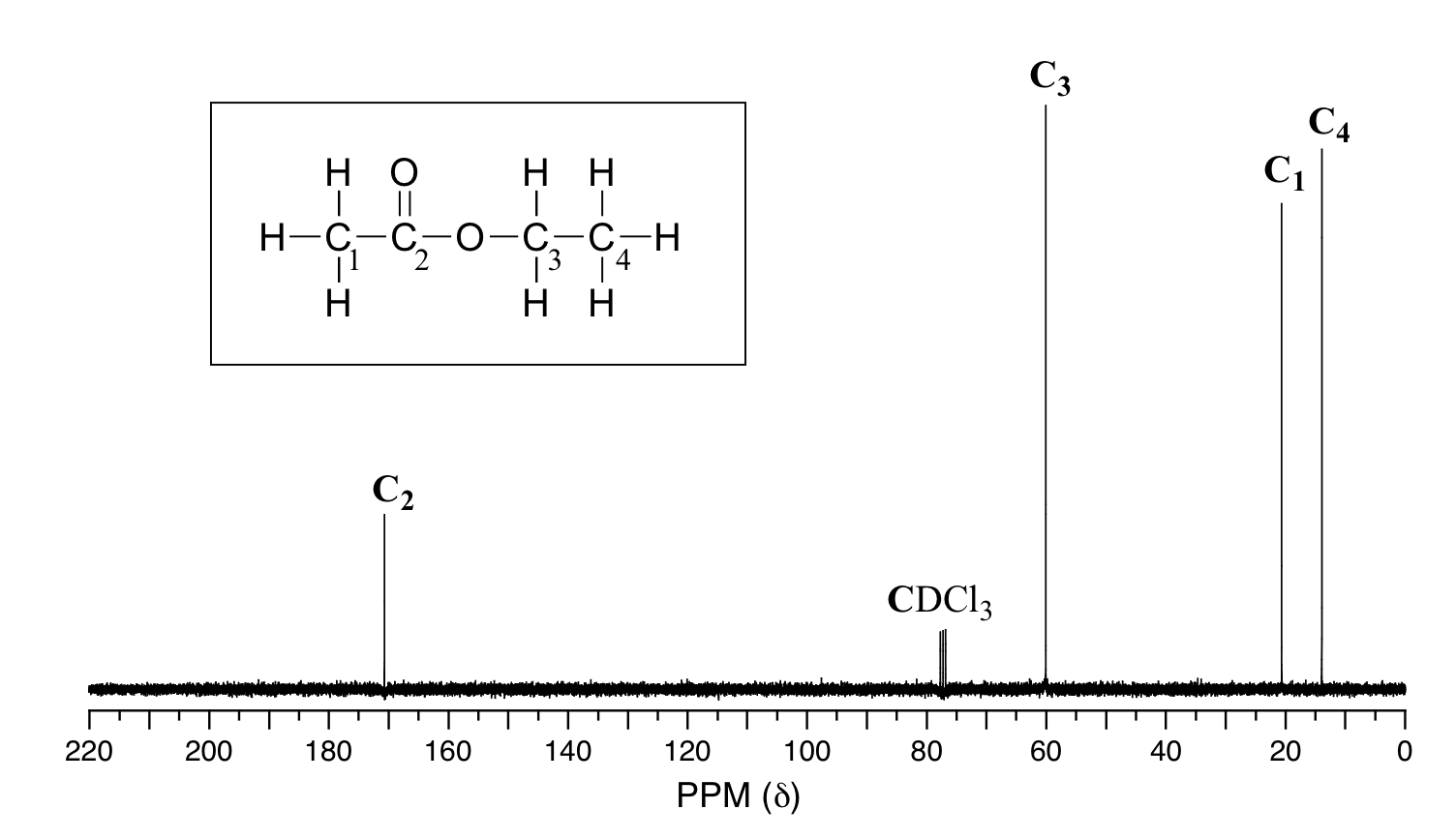
They give the numbers of each type of C atom
(b) Off-Resonance Decoupled Spectra
They give the numbers of each type of C atom and number of H atoms attached to each C.
DEPT —
A technique called DEPT can identify the multiplicity of peaks in an NMR spectrum.
Often you run several different spectra.
The DEPT spectra for an alkyne are shown below.

They show:
- DEPT-135:
#"CH"# and#"CH"_3# peaks up,#"CH"_2# peaks inverted - DEPT-90:
#"CH"# peaks only - "Normal": all
#"C"# atoms
Note that they do not show the quaternary alkyne carbons, as the normal spectra dp.
The DEPT spectra can be combined to yield "edited DEPT" spectra, in which each spectrum contains peaks of only one multiplicity (
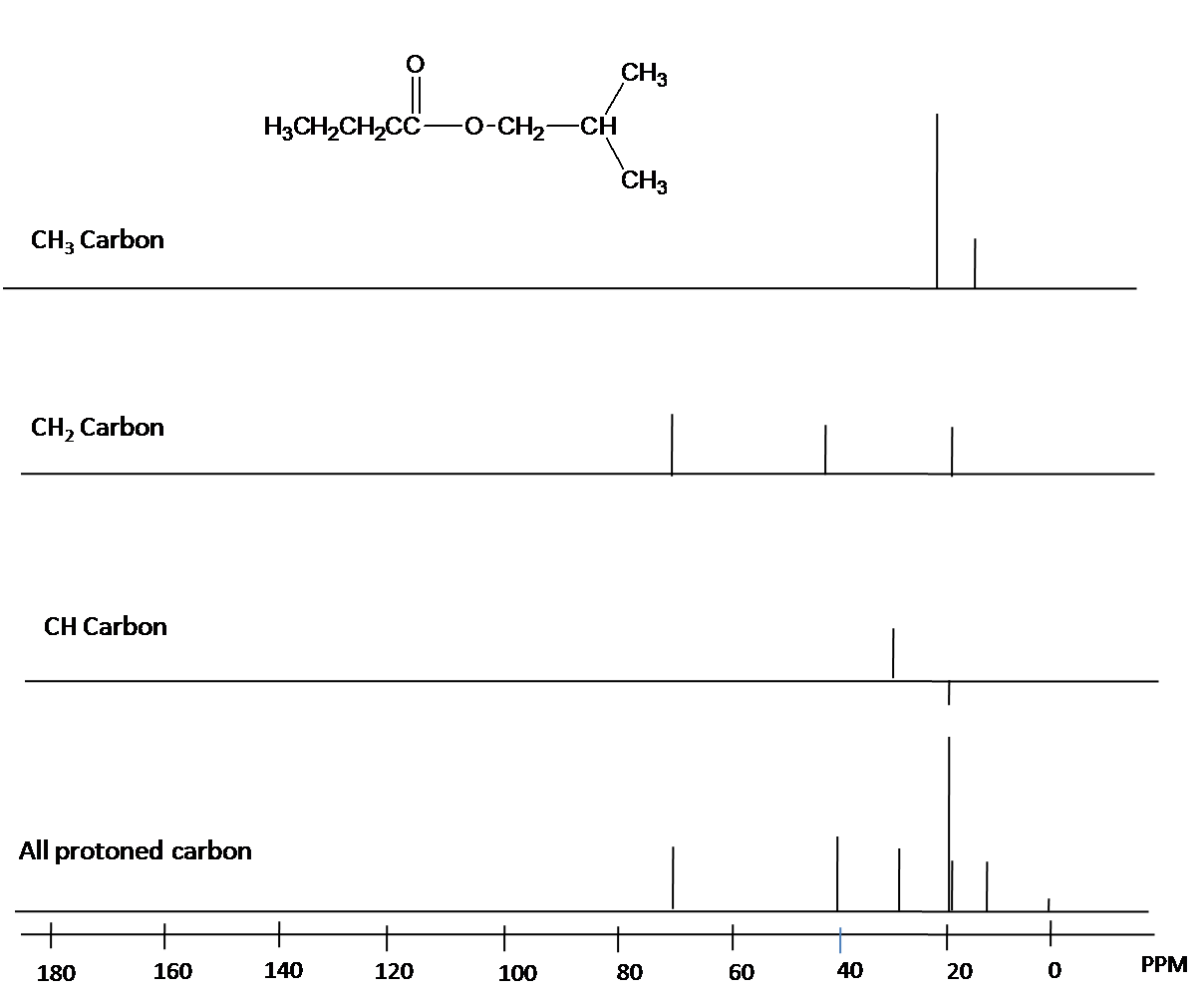
Put this information all together, and you have much information to help you identify a molecule.


