What NMRs Actually Tell Us
Key Questions
-
An NMR spectrum looks like a series of peaks on a graph.
Nuclei like ¹H and ¹³C have a magnetic moment. When you put them in an external magnetic field, they can line up either with or against the field.
If you then apply a radiofrequency field, some nuclei absorb energy and jump to the higher energy level (against the field).
When they return to the lower level, they emit quanta of energy at specific frequencies.
A receiver detects this energy and a computer plots a graph of intensity vs. frequency.
A proton NMR spectrum of diethyl ether looks like this.
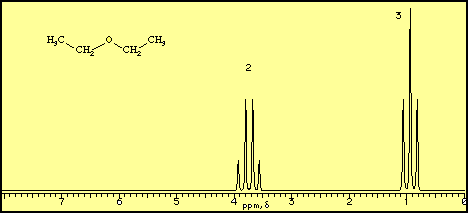
The horizontal axis is in units of δ or ppm "downfield" from a reference point at δ = 0. The vertical axis is in units of relative intensity.
We see signals centred at δ 0.92 and 3.75 with area ratios of 3:2. These correspond to the CH₃ and CH₂ groups in the molecule.
Each signal is split into "multiplets". These give more information on the structure of the molecule.
A ¹³C NMR spectrum of diethyl ether looks like this.

The signals are so narrow that they look like vertical lines, and they are not split into multiplets.
The spectrum tells us that there are two different environments for the carbon atoms in the molecule.
-
Answer:
It is a characterization tool.
Explanation:
NMR is widely used in organic chemistry labs mainly to identify the molecular structure organic molecules, but it could also be used to track the progress of chemical reactions and for many other purposes.
-
Answer:
Proton nuclear magnetic resonance (NMR) spectroscopy exploits the magnetic properties of certain nuclei.
Explanation:
Theory of Nuclear Magnetic Resonance:
A nucleus of an odd atomic/mass number has a nuclear spin that can be observed by the NMR spectrometer. A proton is the simplest nucleus with an odd atomic number of 1, implying it has a spin. We can visualize a spinning proton as a rotating sphere of positive charge. This movement of charge generates a magnetic field, called the magnetic moment, that looks like the field of a small bar magnetic. When a small bar magnet is placed in the field of a larger magnet, it twists to align itself with the field of the larger magnet-a lower energy arrangement than an orientation against the field. The same effect is seen when a proton is placed in an external magnetic field. The proton's magnetic moment to be aligned either with the external field or against the field (by quantum mechanics). The lower-energy state with the proton aligned with the field is called the alpha spin (
#alpha# -spin) state. The higher-energy state with the proton aligned against the external magnetic field is called the beta -spin (#beta# -spin) state.In the absence of an external magnetic field, proton magnetic moments have random orientations. When an external magnetic field is applied, each proton in a sample assumes the
#alpha# or#beta# state.The energy difference between a proton's two spin states is small, but it can be detected by NMR. When a proton interacts with a photon of just the right amount of electromagnetic energy, the proton's spin can flip from
#alpha# to#beta# or vice versa. A nucleus aligned with the field can absorb the energy needed to flip and become aligned against the field. When a nucleus flips its spin, it is said to be in resonance and its absorption of energy is detected by the NMR spectrometer. This is the origin of the term nuclear magnetic resonance.However, protons in chemical compounds are not isolated, they are surrounded by electrons that partially shield them from the external magnetic field. The electrons circulate and generate a small induced magnetic field that opposes the externally applied field. In a molecule, the electron cloud around each nucleus rotates in response to the external field. This induced rotation is a circular current whose magnetic field opposes the external field. The result is that the magnetic field at the nucleus is weaker than the external field, and we say the nucleus is shielded.
A shielded proton requires a stronger field. If all protons were shielded by the same amount, they would all be in resonance at the same combination of frequency and magnetic field. Fortunately, protons in different chemical environments are shielded by different amounts.
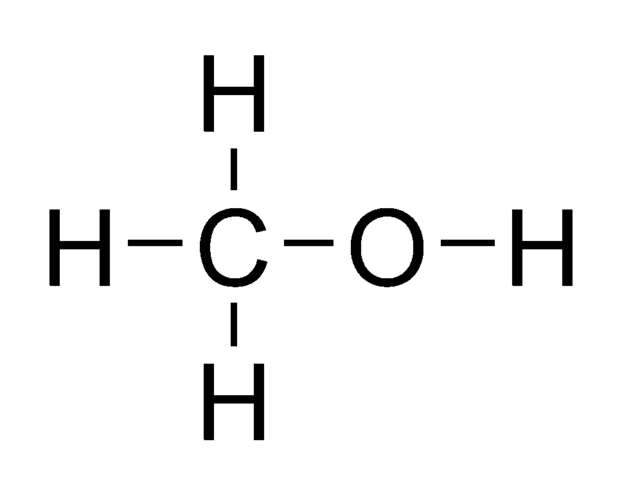
For example, in methanol, the electronegative oxygen atom withdraws electron density from around the hydroxy proton. The hydroxy proton is not shielded as much as the methyl protons, so the hydroxy proton absorbs at a lower field. We say that the hydroxy proton is deshielded by the presence of the electronegative oxygen atom.
NMR spectra provide us with important information:
- The number of different absorptions (signals, peaks) implies how many different types of protons are present.
- The amount of shielding shown by these absorptions implies the electronic structure of the molecule close to each type of proton.
- The intensities of the signals imply how many protons of each type are present.
- The splitting of the signals gives information about other nearby protons.
The NMR Spectrometer
The original, simplest type of NMR spectrometer consisted of four parts:
- A stable magnet with a sensitive controller to produce a precise magnetic field.
- A radio-frequency (RF) transmitter, emitting a precise frequency (continuous wave or CW)
- A detector to measure the same's absorption of RF energy.
- A recorder to plot the output from the detector versus the applied magnetic field.
The printer records a graph of absorption (on the
#y# -axis) as a function of the applied magnetic field (on the#x# -axis). Higher values of the magnetic field are toward the right (upfield) and lower values are toward the left (downfield). The absorptions of more shielded protons appear upfield, toward the right of the spectrum, and more deshielding protons appear downfield, toward the left. Someone else may be able to provide more information on modern NMR spectrometers and how they function.Hope this helps!
Wade, L. G.; Simek, J. W. In Organic Chemistry; Pearson: Glenview, IL, 2013; pp 362–364.