What happens to the atomic mass as you go down each group/family?
1 Answer
As you go down a group, the atomic number increases.
For instance, the noble gases are
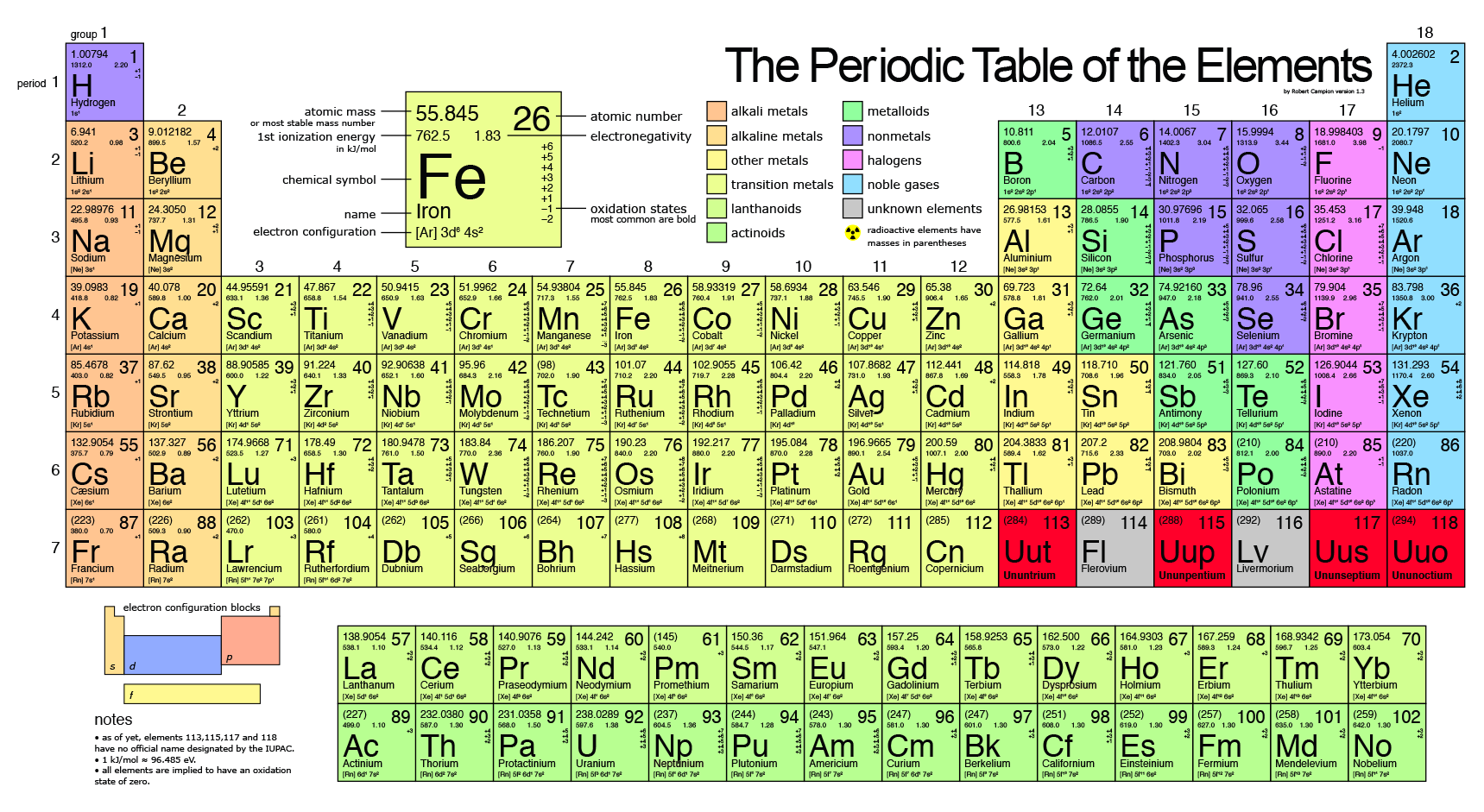
The relative atomic mass is defined as the weighted average of the isotopic masses, while the mass number of each isotope is simply the number of protons plus the number of neutrons, giving each isotopic mass. (So the mass number of
Thus, the relative atomic mass indirectly depends on the number of protons, which directly depends on the atomic number.
So, when the atomic number increases, the mass number of the isotopes is higher (though they are different isotopes), and so the relative atomic mass will increase.
Since this occurs as you go down a group, the atomic mass increases as you go down a group.
When we compare the noble gas relative atomic masses
#M_(r,"He") = 4.0026# #M_(r,"Ne") = 20.1797# #M_(r,"Ar") = 39.948# #M_(r,"Kr") = 83.798# #M_(r,"Xe") = 131.293# #M_(r,"Rn") = ~220#

