What is an example of an enzyme inhibitor?
1 Answer
An enzyme inhibitor is a molecule that binds to enzymes and decreases their activity.
Explanation:
Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors.
The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction.
Enzyme inhibitors also occur naturally and are involved in the regulation of metabolism. For example, enzymes in a metabolic pathway can be inhibited by downstream products.
This type of negative feedback slows the production line when products begin to build up and is an important way to maintain homeostasis in a cell.
Natural poisons are often enzyme inhibitors that have evolved to defend a plant or animal against predators. These natural toxins include some of the most poisonous compounds known.
Artificial inhibitors are often used as drugs, but can also be insecticides such as malathion, herbicides such as glyphosate, or disinfectants such as triclosan.
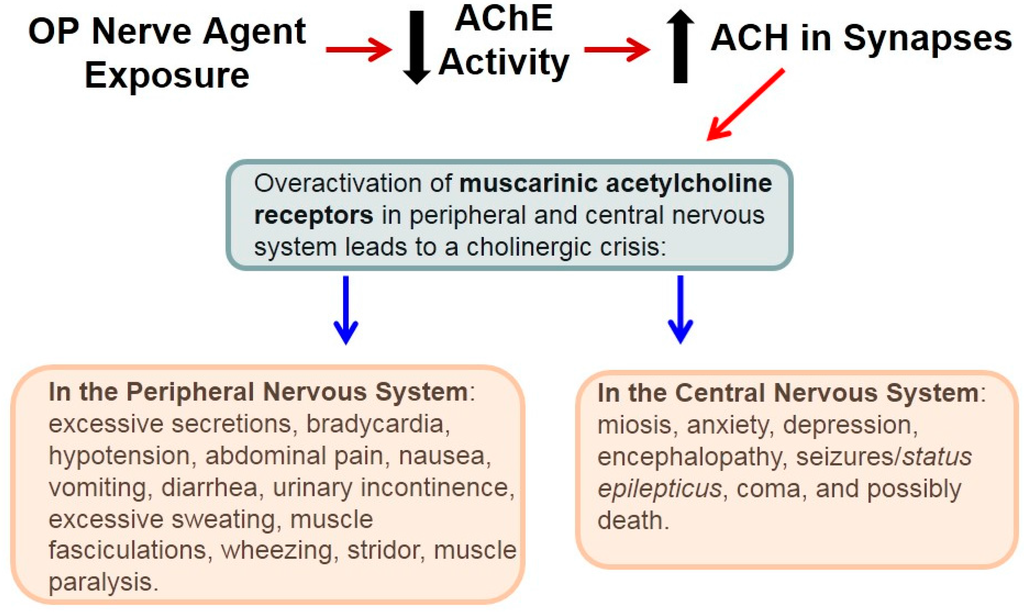
Other artificial enzyme inhibitors block acetylcholinesterase, an enzyme which breaks down acetylcholine, and are used as nerve agents in chemical warfare.
An example of a medicinal enzyme inhibitor is sildenafil (Viagra), a common treatment for male erectile dysfunction.
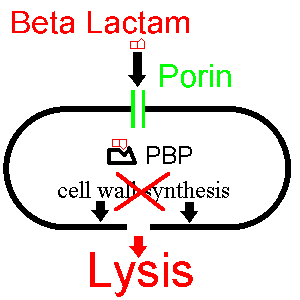
Drugs also are used to inhibit enzymes needed for the survival of pathogens. For example, bacteria are surrounded by a thick cell wall made of a net-like polymer called peptidoglycan.
Many antibiotics such as penicillin and Vancomycin inhibit the enzymes that produce and then cross-link the strands of this polymer together. This causes the cell wall to lose strength and the bacteria to burst.