What is the balanced net ionic equation for sodium hydroxide and acetylsalicylic acid?
1 Answer
Explanation:
Sodium hydroxide and acetylsalicylic acid neutralize each other in a
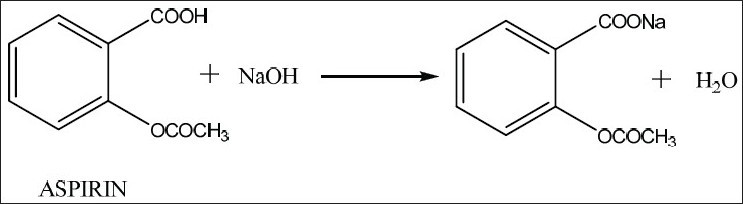
The molecular equation looks like this
#"C"_ 6"H"_ 4 "OCOCH"_ 3"COOH" _ ((aq)) + "NaOH"_ ((aq)) -> "C"_ 6"H"_ 4 "OCOCH"_ 3"COONa"_ ((aq)) + "H"_ 2"O"_ ((l))#
The complete ionic equation looks like this
#"C"_ 6"H"_ 4 "OCOCH"_ 3"COOH" _ ((aq)) + "Na"_ ((aq))^(+) + "OH"_ ((aq))^(-) -> "C"_ 6"H"_ 4 "OCOCH"_ 3"COO" _ ((aq))^(-) + "Na"_ ((aq))^(+) + "H"_ 2"O"_ ((l))#
The sodium cations are spectator ions here, i.e. they are present on both sides of the chemical equation, so you can eliminate them
#"C"_ 6"H"_ 4 "OCOCH"_ 3"COOH" _ ((aq)) + color(red)(cancel(color(black)("Na"_ ((aq))^(+)))) + "OH"_ ((aq))^(-)-> "C"_ 6"H"_ 4 "OCOCH"_ 3"COO" _ ((aq))^(-) + color(red)(cancel(color(black)("Na"_ ((aq))^(+)))) + "H"_ 2"O"_ ((l))#
and write the net ionic equation that describes this reaction.
#"C"_ 6"H"_ 4 "OCOCH"_ 3"COOH" _ ((aq)) + "OH"_ ((aq))^(-)-> "C"_ 6"H"_ 4 "OCOCH"_ 3"COO" _ ((aq))^(-) + "H"_ 2"O"_ ((l))#

