What is the collision theory of chemical reactions?
1 Answer
Jan 1, 2017
The collision theory tells us that a chemical reaction only occurs when particles collide with sufficient kinetic energy.
Explanation:
Besides energy, the orientation of the collision must be right as well.
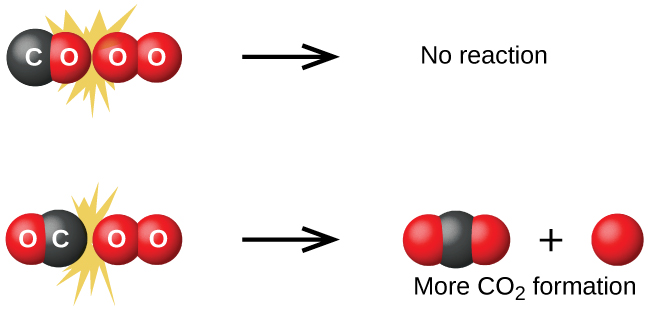
For instance, in the image above, Carbon must collide with oxygen for a reaction to occur.
This video explains it in a clear manner alongside visuals.

