What is the difference between adiabatic process and isothermal process?
1 Answer
May 17, 2018
See Below and see this link for more details.
Explanation:
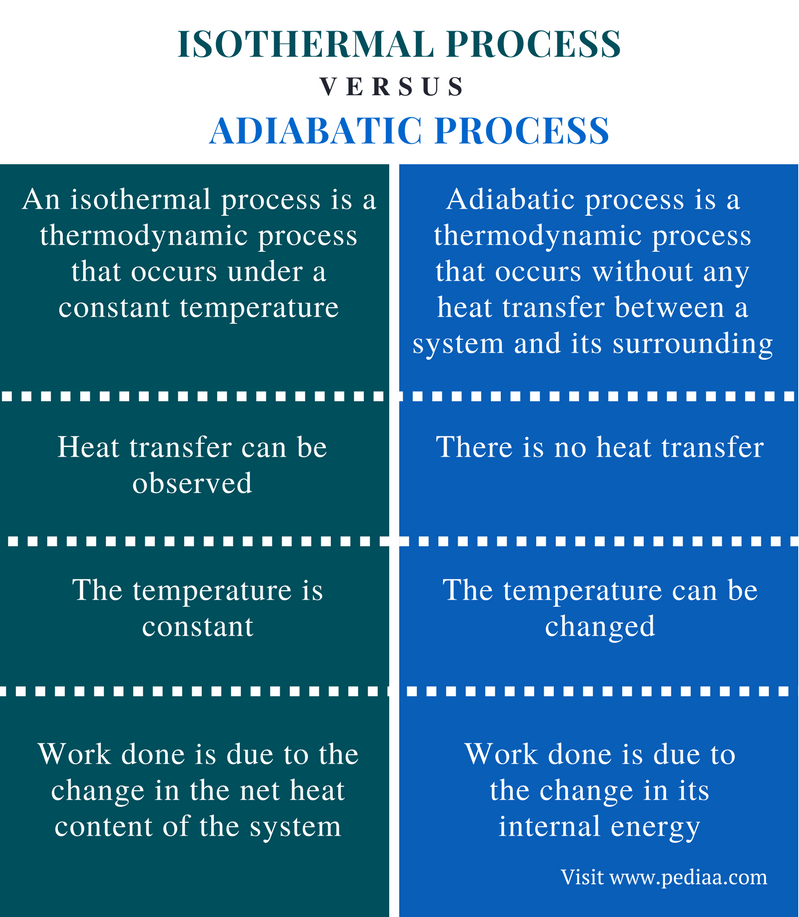
Well, The image says it all. Visit the site link i've provided to know more.
Defintions :
i) Isothermal Process:- Isothermal Process is a change of a system, in which the change in tempeature is zero i.e
And, Of course, this is an ideal process.
ii) Adiabatic Process:- An Adiabatic Process is the change in system that occurs without transfer of heat or a matter between a thermodynamical system or its surroundings; i.e.
Hope this helps.

