What is the difference between an acid and an electrophile?
1 Answer
Although both do accept electrons, there is indeed a difference.
The purpose of an acid is to take electrons in order to donate a proton. The purpose of an electrophile is to take electrons in order to make a significant bond.
Another way to say it is that an acid participates in a chemical reaction "passively" (it stays out of much of the interesting action), whereas an electrophile behaves "actively" (it participates in the interesting action).
The difference can be easily seen here:
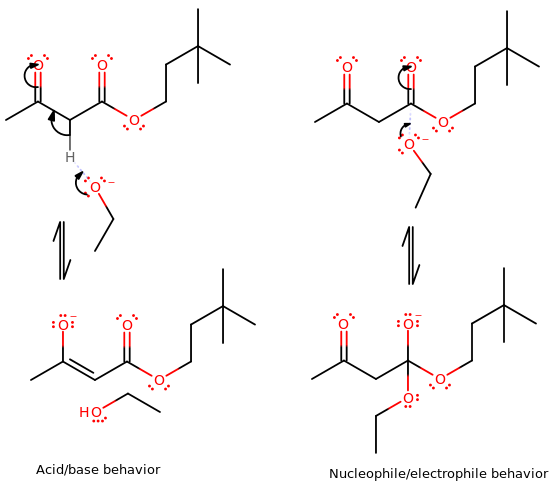
You can see that on the left, the alkoxide wants a proton, but there is no significant bond made between the alkoxide and the big molecule. That is acidic behavior from the big molecule.
But on the right, a significant bond IS made between the alkoxide and the big molecule. That is electrophilic behavior from the big molecule.
(the big ester portion of it to the right is exaggerating that it's supposed to be the leaving group in the mechanism.)

