What is the difference between glucose and glycogen?
1 Answer
Glucose is a monomeric hexose (six-carbon sugar), but when placed into aqueous solution, it isomerizes into its cyclic form. We will focus on its cyclic form because that is where the comparison with glycogen becomes clearer.
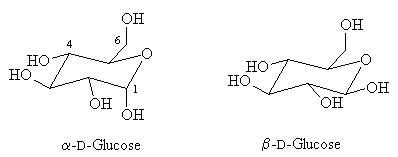 https://www.uic.edu/
https://www.uic.edu/
Glycogen is essentially polymeric glucose. It forms when the protein glycogenin links 10 glucose units together via
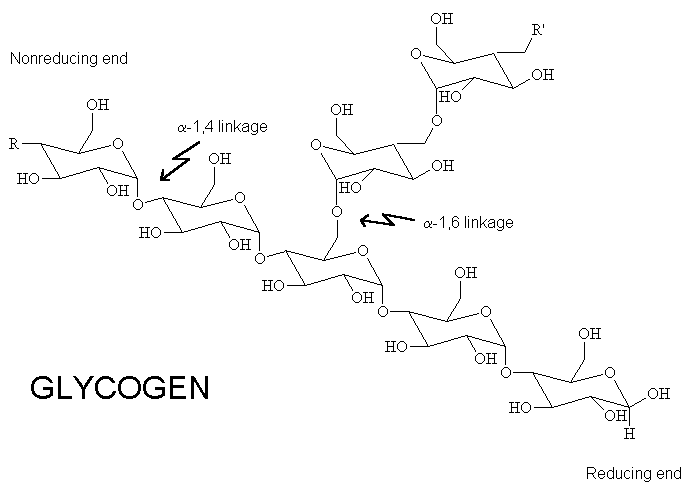 https://www.uic.edu/
https://www.uic.edu/
Afterwards, the enzyme glycogen synthase takes over and catalyzes the linkage of UDP-glucose (uradine diphosphate glucose) with the current form of glycogen (proglycogen) to add more glucose units. This makes a longer glycogen.
(Uradine is composed of a uracil and fructose molecule.)
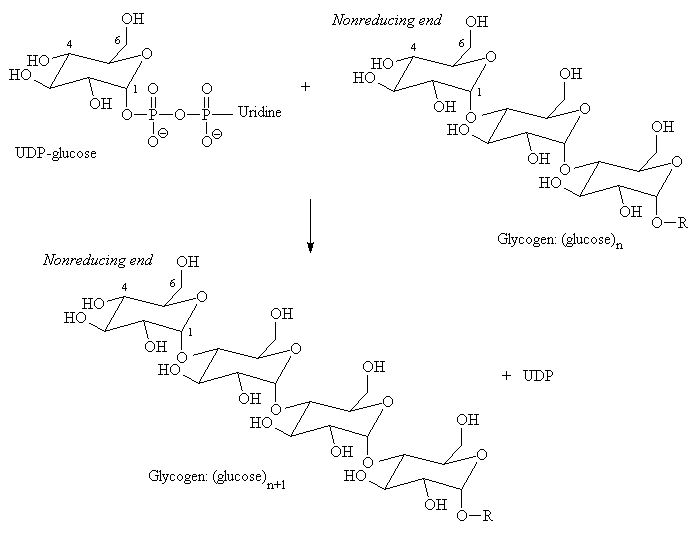 https://www.uic.edu/
https://www.uic.edu/
Glycogen is often used as a source for glucose.
(Note that as you add more glucose units to glycogen, it is still a glycogen. More than 10 units is macroglycogen.)

