What is the difference between structural isomers and geometric isomers? What are examples of both?
1 Answer
Well structural isomers differ in their connectivity..........
Explanation:
And geometric isomers have identical connectivity, but differ in their geometry, their peculiar spatial arrangement.
If we take
On the other hand, for
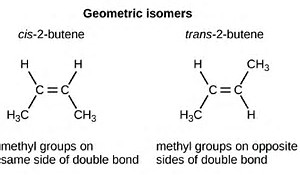
What distinguishes the cis versus trans isomers is the disposition of the alkyl groups across the double bond. This is clearly a geometric difference that results in different physical properties, and different chemistry.
Capisce?

