What is the electron configuration, orbital diagram, and noble gas notation of potassium?
1 Answer
Dec 17, 2014
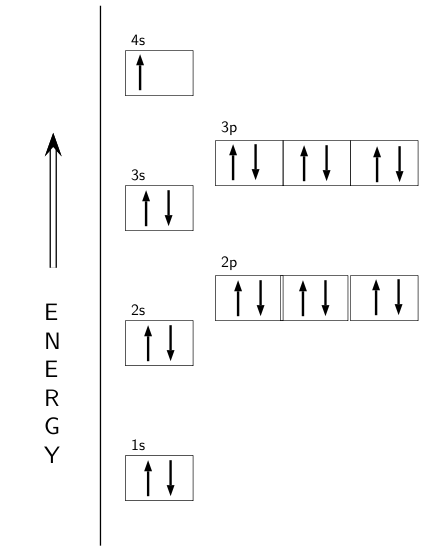
Potassium's atomic number is 19. This means that every atom of potassium has 19 protons in its nucleus. In a neutral atom, the number of protons is equal to the number of electrons. So the electron configuration of potassium will involve 19 electrons.
The full electron configuration of potassium is
The noble gas notation is
The following orbital diagram shows the increase in energy from one energy sublevel to the next, but you can write them on the same level horizontally,