What is the Lewis dot structure for carbon monoxide?
1 Answer
May 22, 2017
See explanation:
Explanation:
Begin finding the chemical formula for carbon monoxide. That is
Then determine how many valence electrons each element has:
Carbon has
I realize that explaining how to draw the lewis dot structure in words might get confusing so what I'm going to do is put a picture of what the diagram will look like along with a video showing step by step on how to exactly create the diagram for this specific example.
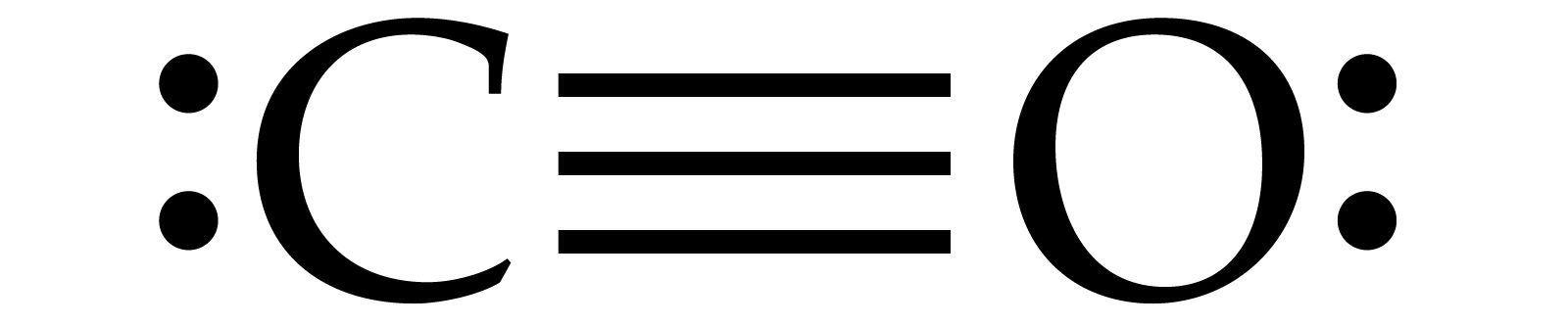
Click this ()

