What is the order of decreasing reactivity toward electrophilic aromatic substitution for compounds: toluene, benzene, fluorobenzene, nitrobenzene, and phenol?
1 Answer
Electrophilic aromatic substitution (EAS) is where benzene acts as a nucleophile to replace a substituent with a new electrophile. That is, benzene needs to donate electrons from inside the ring.
So, benzene becomes less reactive in EAS when deactivating groups are present on it.
Deactivating groups are often good electron-withdrawing groups (EWGs).
These compounds you listed look like this:
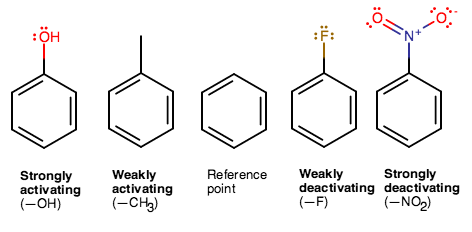
These are, from left to right: phenol, toluene, benzene, fluorobenzene, and nitrobenzene.
From left to right, the amount of electron donation into the ring (activating the ring towards EAS) decreases.
#"OH"# is a#sigma# donor (its HOMO is a#sigma# bonding orbital), thereby activating the ring strongly.#"CH"_3# donates via hyperconjugation (from its#"C"-"H"# bonds), thereby activating the ring weakly.#"F"# is a decent#pi# donor (filled#p# nonbonding orbitals) but is a good EWG, thereby deactivating the ring weakly.#"NO"_2# is a very strong EWG, thereby deactivating the ring strongly.
Therefore, from left to right, we have most to least reactive towards EAS.
Activating groups are favorably ortho/para directors, while deactivating groups are favorably meta directors, with respect to incoming substituents.

