What is the pH of a 0.01 M solution of the strong acid #HNO_3# in water?
1 Answer
Explanation:
The important thing to keep in mind when dealing with strong acids is that they dissociate completely in aqueous solution to form hydronium ions,
Complete dissociation means that every molecule of acid will ionize, i.e. donate its acidic proton to a water molecule.
That tells you that a strong monoprotic acid will produce hydronium ions in a
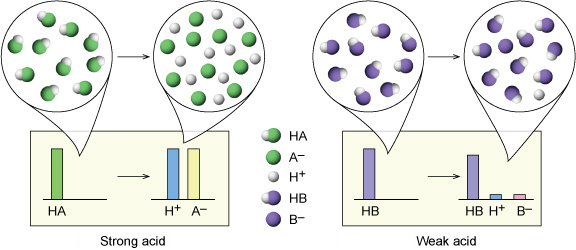
In this case, nitric acid will dissociate to form
#"HNO"_text(3(aq]) + "H"_2"O"_text((l]) -> "H"_3"O"_text((aq])^(+) + "NO"_text(3(aq])^(-)#
This means that the concentration of hydronium ions will be equal to that of the nitric acid
#["H"_3"O"^(+)] = ["HNO"_3] = "0.01 M"#
As you know, a solution's pH is simply a measure of its concentration of hydronium ions
#color(blue)("pH" = - log( ["H"_3"O"^(+)]))#
In this case, the pH of the solution will be
#"pH" = - log(0.01) = color(green)(2)#

