What is the pH of a solution that has a hydronium ion concentration of 0.050 M?
1 Answer
Aug 6, 2016
That solution has a pH of 1.30
Explanation:
The pH can be found using the formula below:
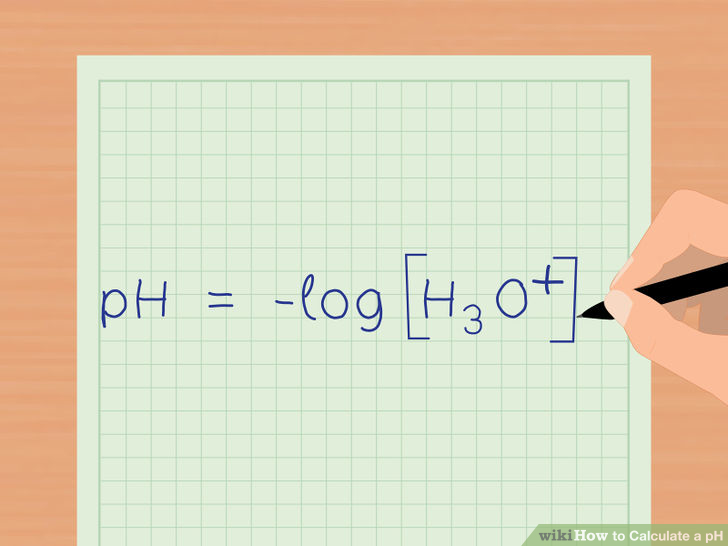
Take the -logarithm of the concentration of hydronium ions that are in the solution:

