What is this equation balanced: Octane reacts with oxygen to form Carbon dioxide and water?
1 Answer
This is the combustion reaction for octane,
The balanced chemical reaction is
Here's a nice diagram of this reaction I've found on a French forum
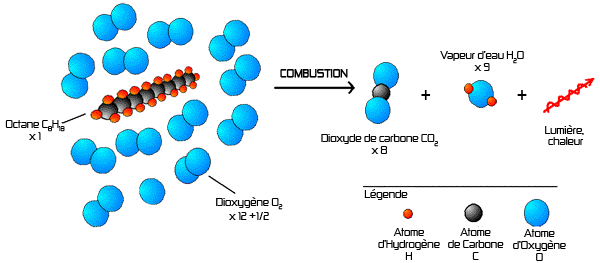
The chemical reaction described is


