What oxidation state is iron in hemoglobin?
1 Answer
Heme in hemoglobin starts out like this:
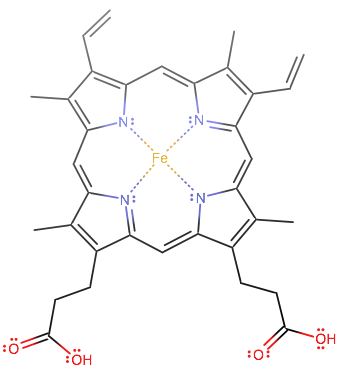
A histidine can bind at the fifth coordination site, while
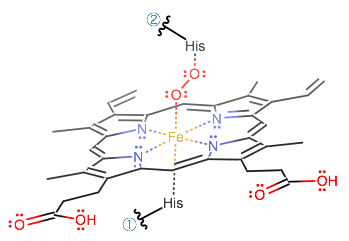
When
#"O"_2 + e^(-) -> "O"_2^(-),#
and the oxidation half-reaction is
#"Fe"^(2+) -> "Fe"^(3+) + e^(-),#
giving the overall reaction as:
#"O"_2 + cancel(e^(-)) -> "O"_2^(-)#
#"Fe"^(2+) -> "Fe"^(3+) + cancel(e^(-))#
#"-----------------------------------------"#
#color(blue)("O"_2 + "Fe"^(2+) -> "Fe"^(3+) + "O"_2^(-))#
On the other hand,

