What types of elements combine with each other will usually result formation of ionic bonding? Give an example.
2 Answers
Usually, metals and non-metals would combine with each other to form an ionic bond.
Explanation:
For example: sodium fluoride
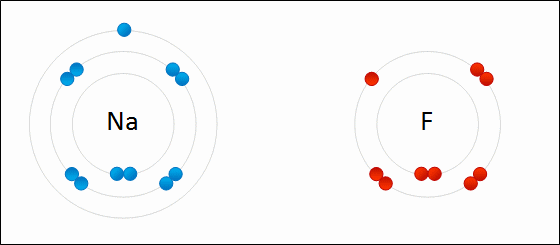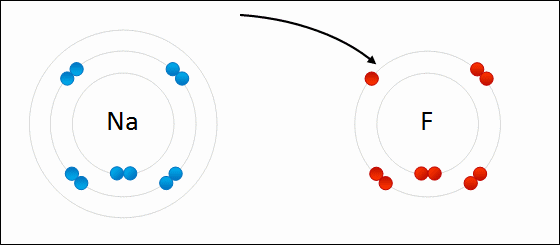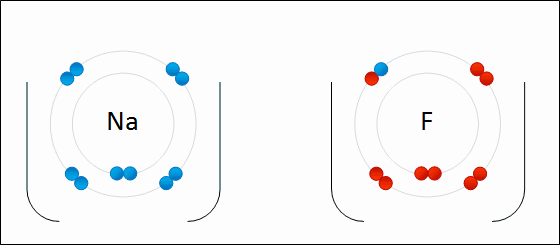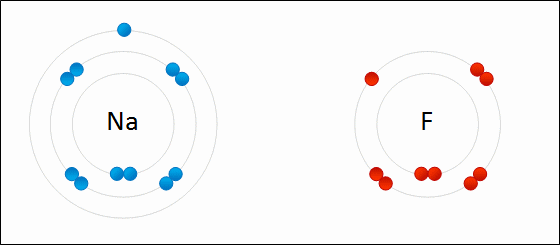
Elements with a low electronegativity combine with elements with a high negativity to form ionic bonding compounds.
Explanation:
An ionic bond is defined by a large difference in electronegativity. The difference in electronegativity causes the electron density to be centered much closer to one element than the other. This causes one element to have a "net" negative charge, while the other element has a "net" positive charge.
The only way to obtain a large difference in electronegativity is for one element ( usually an atom from the left side of the periodic table ) to have a low negativity while the other element ( usually a non metal from the right side of the periodic table) has a high electronegativity.

