What was Einstein's explanation for the photoelectric effect?
1 Answer
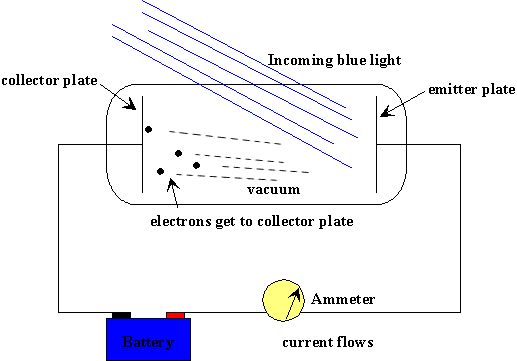
It was observed experimentally that various metals ejected electrons when light of specific or higher frequency was shown on their surface. The wave theory of light could not explain this phenomenon.
Explanation:
In 1900 Max Planck made an ad hoc mathematical assumption of quantized energy to explain Black body radiation.
He proposed the famous expression
Subsequently in 1905 Albert Einstein extended Planck's black body model to propose his solution to explain the photoelectric effect and all other associated observations.
He described light as composed of discrete quanta, now called as photons, as opposed to continuous waves. Einstein extended Planck's assumption to light and proposed that the energy in each quantum of light was equal to the frequency multiplied by a constant. A single photon, whose frequency was above the threshold frequency for a specific metal, has the required energy to eject one electron thus creating the observed photoelectric effect
To explain other experimental results of photoelectric effect, Einstein theorized the following equation:
where

