Which element in the periodic table has the greatest electronegativity? Which has the least electronegativity?
2 Answers
Explanation:
Electronegativity increases across a Period, an horizontal row of the Periodic Table from left to right as we face the Table, and DECREASES down a Group, a column of the Periodic Table.
And thus fluorine has the greatest electronegativity, which is consistent with its status as the most reactive element on the Periodic Table. See here.
And it follows that the lower alkali metals are least electronegative........
Highest:
Lowest:
Explanation:
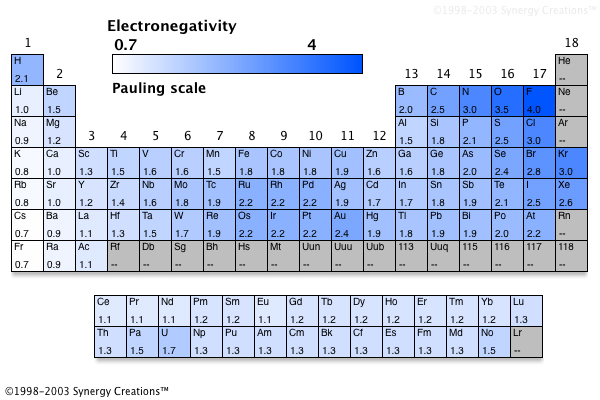
This table gives us a color-coded visualization of the trends for electronegativity on the periodic table. We can see that the lowest electronegativity values are near the bottom-left of the table, and the highest values are near the top-right.
From this, we can see that both cesium (
Why is this the trend?
In simple terms, electronegativity is a measure of the ability of an atom to pull or attract electrons toward itself when it is chemically bonded to another element. The higher the electronegativity, the more that atom's nucleus "pulls" the bonding electrons toward itself.
We can generalize the fact that if an atom is larger, the farther the electrons are located relative to the nucleus, and the weaker the nucleus is able to pull electrons toward itself. In contrast, the smaller the atom, the closer the electrons are to the nucleus, and the nucleus can pull them in with a stronger force.
Therefore, electronegativity is (most generally) inversely proportional to atomic radius. That is, the larger the radius (larger the size), the lower the electronegativity.
That's why in the diagram the larger atoms (near the bottom-left of the table) have a lower electronegativity, and the smaller atoms (near the top-right) have a higher electronegativity. Fluorine and francium happen to be the extreme corners of the table (noble gases generally aren't given electronegatvity values, due to their general lack of chemical reactivity).

