Which substance is oxidized ad which is reduced in the following redox reactions?
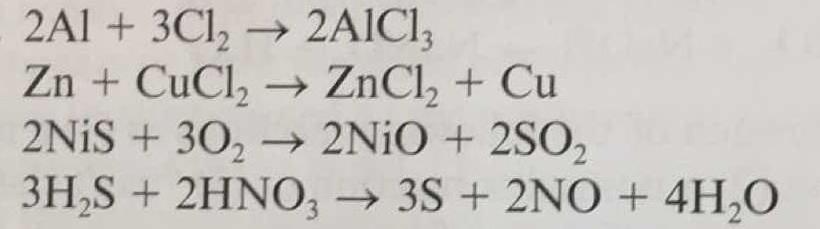
1 Answer
The reduction reaction is the one involving an electron being gained
The oxidation reaction involves an electron being lost
So, the metal is usually oxidised, because metal only have 1 or 2 electrons in their outer shell so they are keen to lose them.
Non-metals, especially halides (group 7) will be reduced because they have 6 or 7 electrons in their outer shell, so want to gain a few.
It is worth learning your ions for this, as am atom that forms a negative ion will be the one being reduced (and vice versa).
So, for the examples above:
Aluminium is oxidised, Chloride is reduced
Zinc is oxidised, chloride is reduced, copper is displaced (kicked out by a more reactive metal)
i'm sure you've noticed the next one is a bit trickier...
I think it's to do with the oxygen being covalently bonded, so there are no ions. Then, the oxygen becomes a 2- ion so it can bind with the nickel, so it has gained electrons and been reduced.
The nickel stays as an ion the whole time, so it's a spectator ion.
The sulfur changes from an ion to an atom so it can covalently bond with the oxygen, so to go from a 2- ion to a neutral atom it must have lost electrons, so the sulfur is oxidised.
The last one I'm afraid I'm not sure about, so I'm going to ask about it today and get back to you! I thought it would be better to help with the ones I could first.
