Why are alpha particles positive?
1 Answer
Dec 29, 2014
An alpha particles is positively charged because it is essentially the nucleus of a Helium-4 atom.
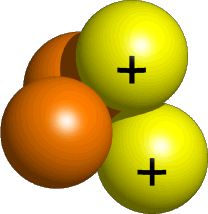
A Helium-4 nucleus is composed of two protons, which are positively charged particles, and two neutrons, which have no electric charge.
A neutral He atom has a mass of four units (2 protons + 2 neutrons) and a net charge of zero because it has two electrons that balance the protons' positive charge; since an

