Why is Boyle's law graph curved?
1 Answer
Boyle's law expressed the inverse relationship between an ideal gas' pressure and its volume if temperature is kept constant, i.e. when pressure increases, volume decreases, and vice versa.
I won't detail how to graph this relationship, since it has been answered in great detail here:
http://socratic.org/questions/how-do-you-graph-boyles-law?source=search
Now, here's how the
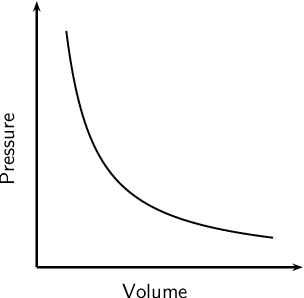
If you were to do an experiment and plot the
The interesting thing about a hyperbola is that it has two asymptotes, a horizontal one and a vertical one. An asymptote is essentially a line that a curve approaches as it heads to infinity.
The physical explanaition for the existance of these asymptotes is the fact that no matter how much the pressure increases, the volume can never be zero; likewise, pressure can never be zero as this would mean an Infinitely large volume.
In other words, you would need infinite pressure to compress a gas completely. Likewise, pressure can never be zero because, theoretically, the gas would expand to an infinite volume.
So, even without having any experimental data to fit into a graph, one could estimate that the inverse relationship between pressure and volume must have two asymptotes, and if that's the case, it must be a curve.

