Boyle's Law
Key Questions
-
Boyle's Law is a relationship between pressure and volume at a constant temperature.
P_1V_1 = P_2V_2 In this relationship, pressure and volume have an inverse relationship when temperature is held constant.
If there is a decrease in the volume there is less space for molecules to move and therefore they collide more often, increasing the pressure. If there is an increase in the volume the molecules have more space to move, collisions happen less often and the pressure is decreased.
vV ^P ^V vP the relationship is inverse.
However if temperature is involved the relationship between the three values is the combined gas law.
(PV)/T = (PV)/T I hope this was helpful.
SMARTERTEACHER
-
Boyle's law describes how the pressure of a gas tends to decrease as the volume of a gas increases.
Boyle's Law
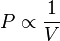
When the Volume Decreases, the Pressure increases.

(mass and temperature is constant)
