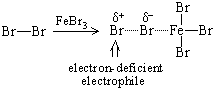As you probably know, a Lewis acid is a compound that is capable of accepting electron pairs.
If you look at #FeBr_3#, the first thing that should stand out is the fact that you've got a transition metal, #Fe#, bonded to a highly electronegative element, #Br#.
This difference in electronegativity creates a partial positive charge on the #Fe#, which in turns allows it to accept an electron pair. Remember that transition metals are capable of expanding their octets in order to accomodate more electrons, so a good rule of thumb is that compounds formed by transition metals paired with highly electronegative elements will most likely be Lewis acids.
#FeBr_3# is used as a catalyst in the bromination of benzene because of its strong Lewis acid character.
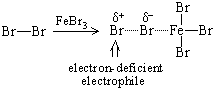
Without going into detail, the bromine (#Br_2#) molecule reacts with #FeBr_3# and donates a pair of electrons to it. This polarizes #Br_2#, making it a better electrophile (more attracted to electrons).