Why is Fischer esterification not useful for tertiary alcohols?
1 Answer
Dec 23, 2015
Fischer esterification not useful for tertiary alcohols because the strong acid catalyst causes them to dehydrate.
Explanation:
Fischer esterification is the acid-catalyzed reaction between an alcohol and a carboxylic acid to form an ester.

The first step in the mechanism is protonation of the carbonyl group.
This enhances its susceptibility to attack by the alcohol.
Of course, the alcohol is also protonated by the acid catalyst.
A tertiary alcohol is easily dehydrated by the strong acid catalyst in an
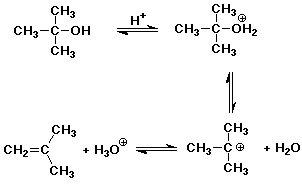
The dehydration is usually much faster than the esterification.
Therefore, Fischer esterification is not useful for tertiary alcohols.

