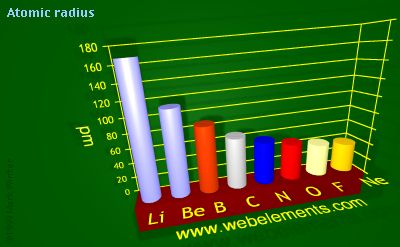
Why is the atomic radius of Li larger than that of Be?
1 Answer
Here's what's going on here.
Explanation:
Lithium,
For both these elements, the outermost electrons are located on the second energy level in the 2s-subshell. However, these outermost electrons. which ultimately determine the atomic radius, are closer to the nucleus in beryllium's case.
That happens because beryllium has a higher effective nuclear charge,
The effective nuclear charge is simply a measure of the net positive charge that affects the outermost electrons in an atom.
In lithium's case, the outermost electron is being screened from the nucleus by two core electrons. The same is true for the two outermost electrons in beryllium, they are screened by two core electrons.
However, beryllium has
This means that the nucleus of a beryllium atom will pull on the outermost electrons with more force, effectively compressing the distance between itself and these electrons.
That is why lithium has a larger atomic radius than beryllium. In fact, atomic radius decreases as you move from left to right across a period.