Why is the phase diagram of water so odd?
1 Answer
Because the slope of the liquid-solid coexistence curve (the line
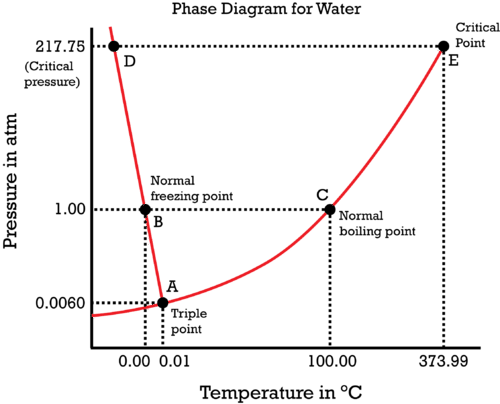
To rationalize this, we turn to the Clapeyron equation.
#(dP)/(dT) = (Delta_fbarH_A)/(T_(f)DeltabarV_A^((s) -> (l)))#
This equation describes the slope of the liquid-solid coexistence curve. Now, since:
- this slope,
#(dP)/(dT)# , is known to be negative, - temperature in
#"K"# is always positive, - and the
#Delta_fH# (enthalpy of fusion) for melting is positive,
the change in molar volume for a melting solvent,
See below if you want to see how one can derive this equation.
DISCLAIMER: This is a derivation. If you have to know it, or if you're curious, read on.
At the liquid-solid coexistence curve, the chemical potential,
#mu_A^((s)) = mu_A^((l))#
Chemical potential is basically the tendency to move towards a certain energetic direction.
Since these chemical potentials are equal, there is no tendency to stay in one particular phase over the other, so the infinitesimally small change in chemical potential for melting is
#dmu_A^((s) -> (l)) ~~ Deltamu_A^((s) -> (l)) = mu_A^((s)) - mu_A^((l)) = 0#
Now, note that
#dG = -SdT + VdP# where
#S# is entropy and the remaining variables (#T,P# ) are well-known.
Similarly, if we let
#dDeltamu_A = dDeltabarG_A = -DeltabarS_AdT + DeltabarV_AdP#
Now, if we set
#0 = -Delta_fbarS_AdT + DeltabarV_A^((s) -> (l))dP#
#Delta_fbarS_AdT = DeltabarV_A^((s) -> (l))dP#
#(Delta_fbarS_A)/(DeltabarV_A^((s) -> (l)))dT = dP#
This gives us the Clapeyron equation:
#color(green)((dP)/(dT) = (Delta_fbarS_A)/(DeltabarV_A^((s) -> (l))))# where
#Delta_fbarS# is the molar enthalpy of fusion. Or, if we recognize that#DeltaG = 0# at a phase equilibrium, then#Delta_fS = (Delta_fH)/T# , and we have an easier version to interpret:
#color(blue)((dP)/(dT) = (Delta_fbarH_A)/(T_(f)DeltabarV_A^((s) -> (l))))# where
#T_f# is the freezing point.
This equation describes the slope of the liquid-solid coexistence curve.

