Why some gases are collected over the water?? Reasons please
1 Answer
Because displacement of water is a convenient means to collect a gas........
Explanation:
In the chemistry laboratory, we often fill a graduated cylinder completely with water, and then invert this into another large, filled beaker, such that the cylinder remains full of water.
A hose is passed to bleed into the open end of the cylinder, and the other end of the hose is attached to your reaction vessel, in which some gas is generated.
The evolved gas displaces some volume of the water, and when the levels of the volumes are equilibrated (i.e. the level of water inside the cylinder is equal to the level of water in the beaker), the pressure inside the cylinder is due to......
...where
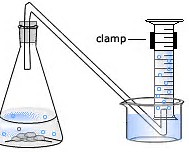
Note that you could collect some gases this way; for instance dioxygen, or dinitrogen, or dihydrogen, or carbon dioxide. You could not collect a gas with substantial water solubility, for instance

