Zirconium has an atomic number of 40. How many neutrons are there in an isotope of zirconium-92?
1 Answer
Mar 30, 2017
52
Explanation:
To determine the number of neutrons that zirconium-92 has we will use the formula below:
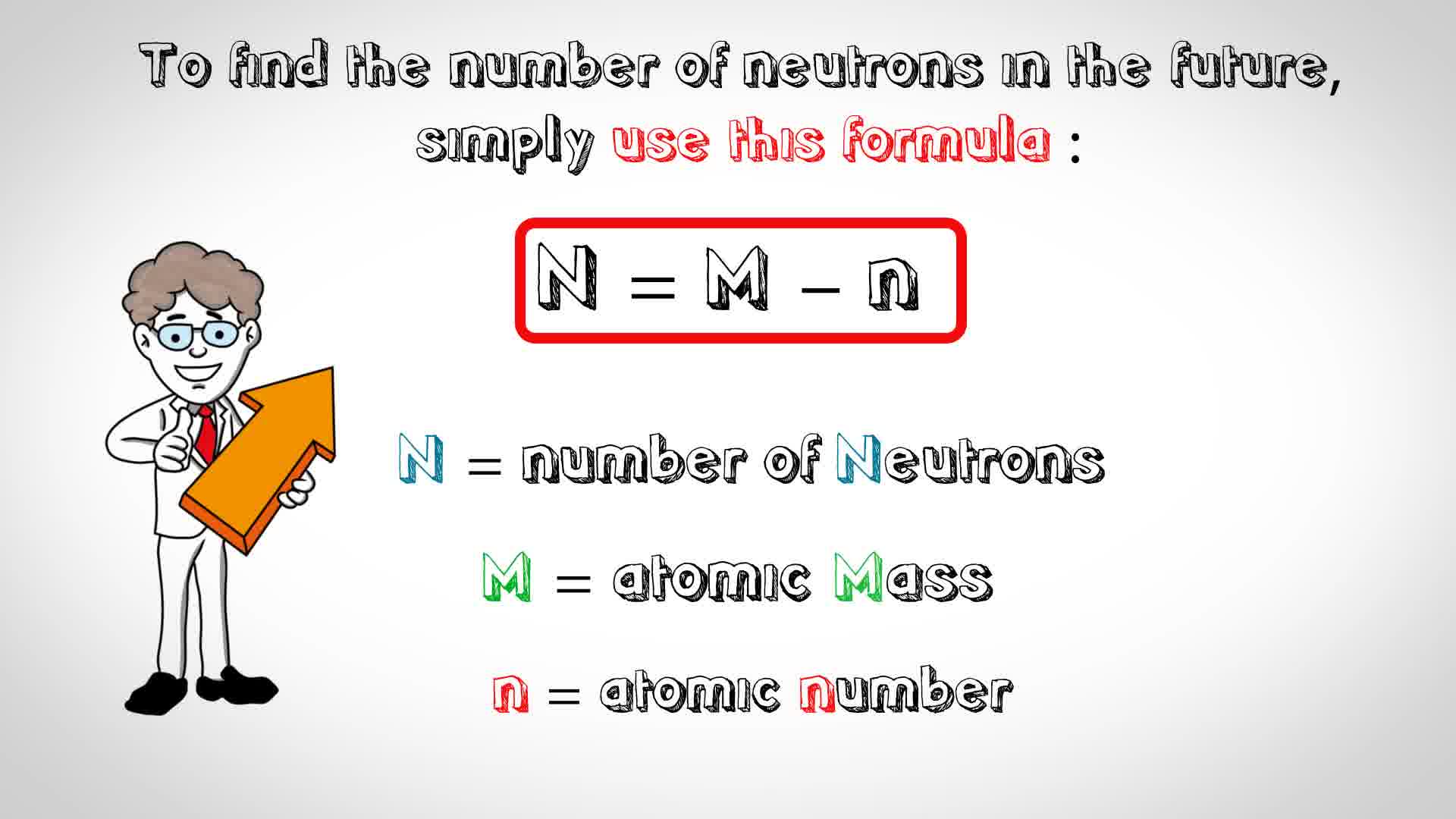
We know the atomic number (40) and we know the atomic mass (92), so all we have to do is plug the numbers into the equation and solve for N:
There are 52 neutrons in the isotope Zr-92
*I should also mention that the number to the right of the element's name is the atomic mass.
I hope this helps!

