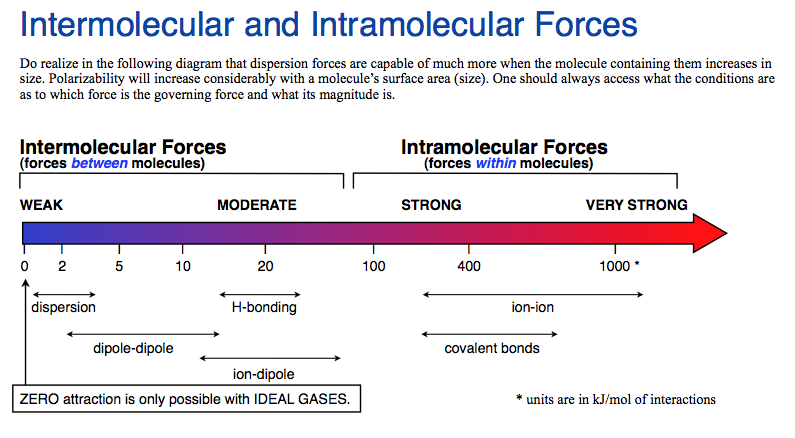
Which of the forces of molecular attraction is the weakest: hydrogen bond, dipole interaction, dispersion, polar bond?
1 Answer
Generally speaking, dispersion forces are the weakest.
Hydrogen bonds, dipole interactions and polar bonds are all based on electrostatic interactions between permanent charges or dipoles.
However, dispersion forces are based on transient interactions in which a momentary fluctuation in the electron cloud on one atom or molecule is matched by an opposite momentary fluctuation on the other, thereby creating a momentary attractive interaction between two mutually induced dipoles.
This attractive dispersion force between two nominally uncharged and unpolarized (but polarizable) atoms is the result of electron correlation between the two atoms. Because the forces are based on momentary and transient interactions, they are exceptionally weak.
Don't forget to check this also