Which set of quantum numbers is allowed?
1) #(n,l,m_l,m_s) = (6,3,-3,+1/2)#
2) #(n,l,m_l,m_s) = (2,2,1,+1/2)#
3) #(n,l,m_l,m_s) = (2,1,-1,-1/2)#
4) #(n,l,m_l,m_s) = (4,2,1,0)#
5) #(n,l,m_l,m_s) = (2,1,-1/2,+1/2)#
6) #(n,l,m_l,m_s) = (0,1,-1,-1/2)#
1)
2)
3)
4)
5)
6)
1 Answer
The correct sets are (1) and (3).
You can think of quantum numbers as being coordinates used to describe the position and spin of an electron in an atom.
There are four quantum numbers, each with its own set of acceptable values
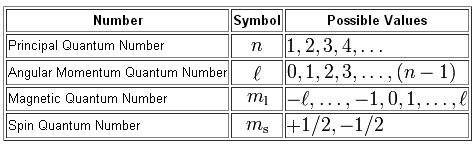
The spin quantum number,
The angular momentum quantum number,
This being said, let's start with the given sets
- Set (1)
All the quantum numbers have allowed values. This particular set describes an electron located in the 6th energy level, in one of the seven 6f-orbitals, and having spin-up.
- Set (2)
Notice that this set has
- Set (3)
Once again, all the quantum numbers have allowed values. This set describes an electron located in the 2nd energy level, in the
- Set (4)
This set is not allowed because
- Set (5)
This time, the value of the magnetic quantum number is not allowed. Notice that
- Set (6)
This set is not allowed because

