What is the Heisenberg uncertainty principle? How does a Bohr atom violate the uncertainty principle?
2 Answers
Basically Heisenberg tells us that you cannot know with absolute certainty simultaneously both the position and momentum of a particle.
Explanation:
This principle is quite tough to understand in macroscopic terms where you can see, say, a car and determine its velocity.
In terms of a microscopic particle the problem is that the distinction between particle and wave becomes quite fuzzy!
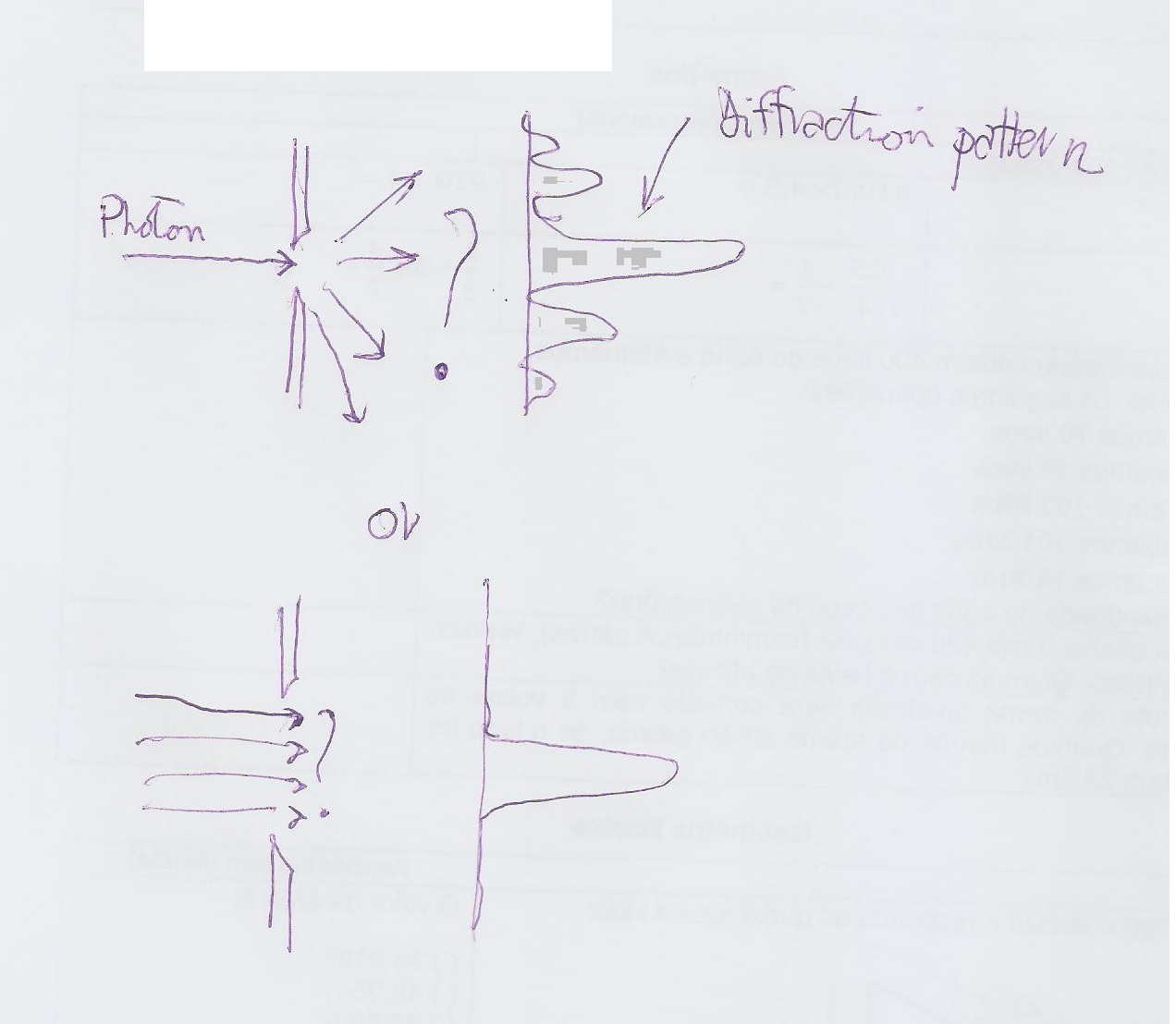
Consider one of these entities: a photon of light passing through a slit.
Normally you'll get a diffraction pattern but if you consider a single photon....you have a problem;
If you reduce the width of the slit the diffraction pattern increases its complexity creating a series of maxima. In this case you can "select" one photon and so its position (at the slit exactly) making the slit very narrow BUT then what will be its momentum? It will even have 2 components (gong in "diagonal")!!!!
If you make the slit very large all the photons will land at the center with the same velocity and so same momentum BUT now which is which???
Bohr's model probably violate the principle because with it you can simultaneously localize the electron (at a certain radial distance) and determine its velocity (from quantization of angular momentum
Hope it is not too confusing!
Heisenberg's uncertainty principle states that you can't know position or momentum exactly, which is what Bohr's model of the atom is based on.
Explanation:
Heisenberg's Uncertainty Principle says that you can't know some properties exactly, such as energy, time taken, position or momentum, at the quantum level.
This is strange, because classical physics (Newton's laws and so on) is built out of definite values, everything acting normally. In quantum physics, this is not so.
When you get to a small enough level - electrons, photons, quarks - things stop acting like particles and golf balls, but instead act a little more like waves. These quantum dots are not in one particular place, like a golf ball, but have a probability density, which means they are probably over here, but could be somewhere else - we can't know exactly.
Bohr's model of the atom is all built out of things acting like golf balls. It has a nucleus very precisely in the centre, and electrons in nice, neat orbitals around the outside, perfect circles with electrons moving around like planets.
Heisenberg's uncertainty introduces to us a different concept entirely. Rather than being in circular orbit, electrons are in fuzzy areas of probability around the nucleus, called orbitals. Orbitals can be circular, too, but some of them are shaped like rings or hour-glasses, and orientated along different axes - nothing like Bohr's shells.


