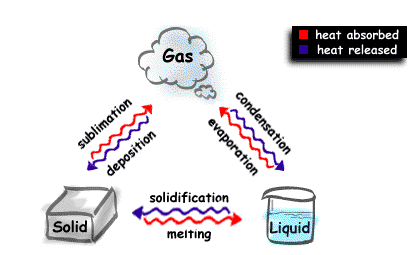
For the three states of matter (solid, liquid, and gas) there are six possible changes of state. Which changes of state are exothermic and which are endothermic ?
1 Answer
Here's what I got.
Explanation:
Any phase change that takes you from higher energy particles to lower energy particles is exothermic.
Similarly, any phase change that takes you from lower energy particles to higher energy particles is endothermic.
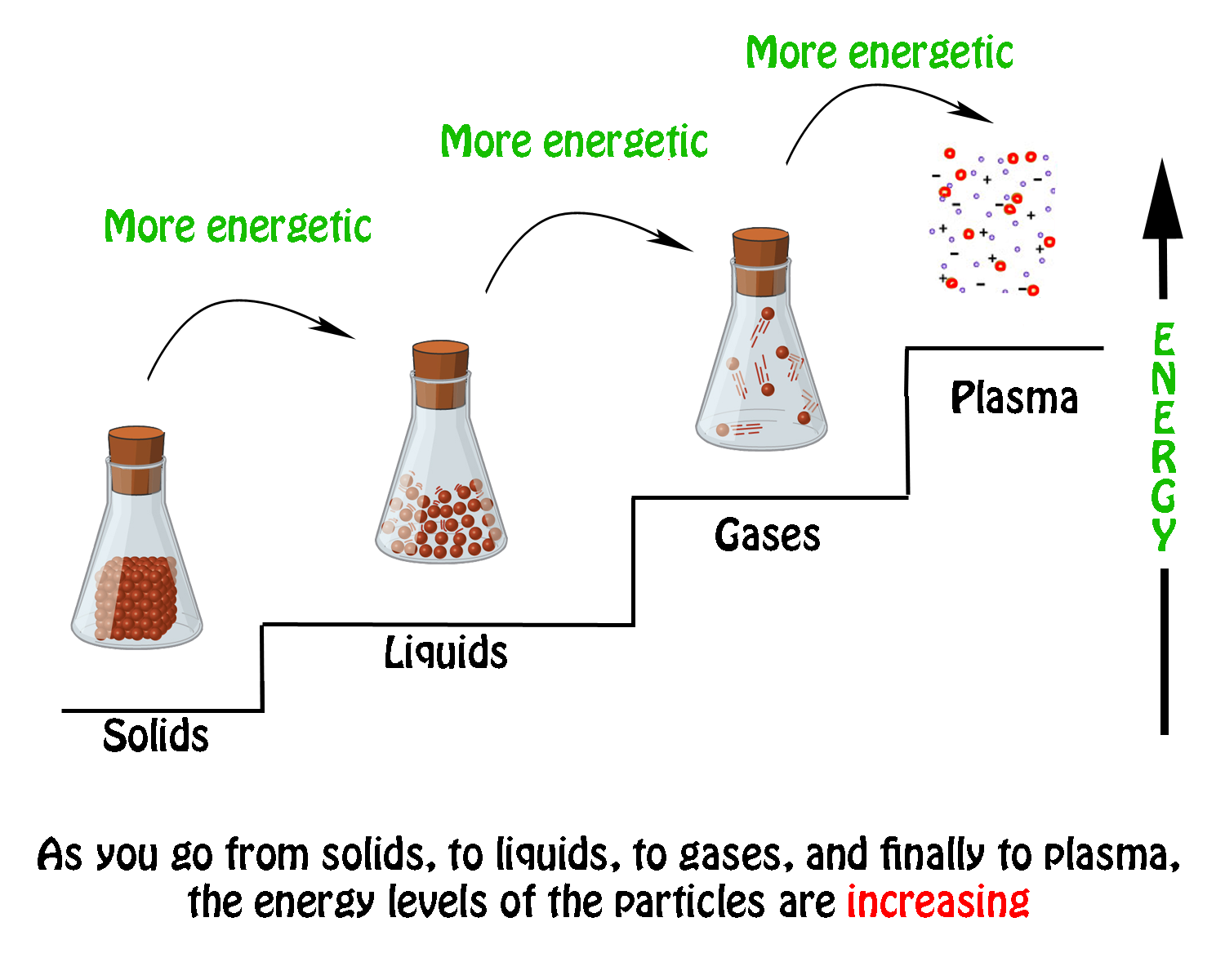
The idea here is that in order to change the state of a substance you must either provide with energy or take away energy.
When you're going from particles that are in a lower energetic state to particles that are in a higher energetic state, you must provide energy.
This implies that the process will be endothermic, since heat must be absorbed. You will thus have -- I'll ignore plasma here and stick to the traditional phase changes
- solid to liquid
#-> color(red)("melting") -> color(red)("requires") color(white)(a)"energy"# - liquid to gas
#-> color(red)("evaporation") -> color(red)("requires") color(white)(a)"energy"# - solid to gas
#-> color(red)("sublimation") -> color(red)("requires") color(white)(a)"energy"#
When you're going from particles that are in a higher energy state to particles that are in a lower energy state, you must take away energy.
This implies that the process will be exothermic, since heat is being released. You will thus have
- gas to liquid
#-> color(blue)("condensation") -> color(blue)("gives off") color(white)(a)"energy"# - liquid to solid
#-> color(blue)("freezing") -> color(blue)("gives off") color(white)(a)"energy"# - gas to solid
#-> color(blue)("deposition") -> color(blue)("gives off") color(white)(a)"energy"#
And there you have it -- six phase changes, three exothermic and three endothermic, correspond to the three traditional phases of matter, liquid, solid, and gas.