Question #81671
1 Answer
The hydrogen bonds between the nitrogenous base pairs of DNA are strong dipole-dipole attractions. They hold the two strands of DNA together.
Explanation:
The nitrogenous bases in DNA are A (adenine), C (cytosine), G (guanidine), and T (thymine). Here are their structures:

In each of these bases, the nitrogen of the secondary amine bonds to the deoxyribose in the backbone.
DNA consists of two strands running in opposite directions. The hydrophobic nitrogenous bases are on the inside, hidden from water.
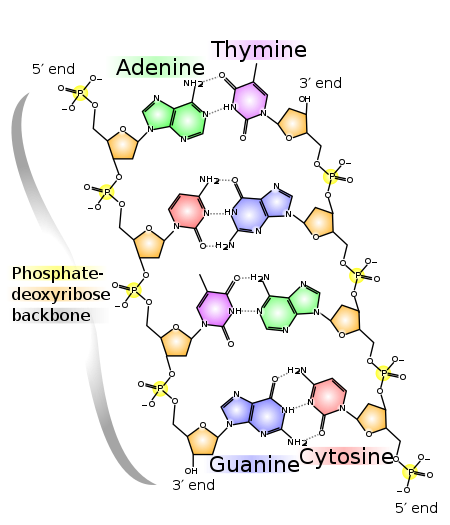
Hydrogen bonds between the nitrogenous base hold the two strands of DNA together. These hydrogen bonds are base specific. That is, A (adenine) can form hydrogen bonds only with T (thymine). C (cytosine) can form hydrogen bonds only with G (guanine).
The thymine-adenine pair interacts through two hydrogen bonds. The cytosine-guanine pair interacts through three hydrogen bonds.

