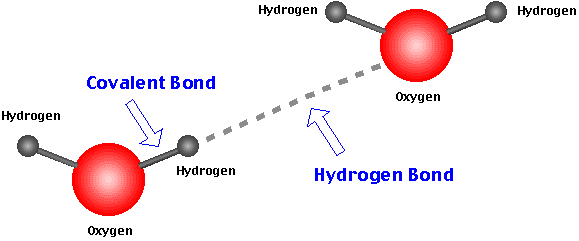
How are hydrogen bonds related to cohesion?
1 Answer
Cohesion means that molecules of a substance are going to stick to each other.
Vapours condense by reducing the temperature or by increasing the pressure. These phenomena are possible thanks to cohesion between molecules.
Molecular solids that have stronger attractions between molecules are harder and higher melting.
Every intermolecular force (IMF) can give similar attractive effects, and a hydrogen bond is one of these intermolecular bonds .
Example I: Cellulose molecules are attracted to each other by many hydrogen bonds, yielding a stiff material such as wood.
Example II: Glycerol is a syrup-like liquid because its molecules can form three or more hydrogen bonds per molecule.
Counter-example: Olive oil is more viscous than water, but its molecules haven't any hydrogen bonds. They stick to each other by means of a different kind of *intermolecular force * .