Question #4abaa
1 Answer
Aug 31, 2015
An atom will be radioactive, if its nuclei is unstable.
Explanation:
-
In an unstable atom, the nucleus changes by giving off particles or energy to get back to a balanced state.
-
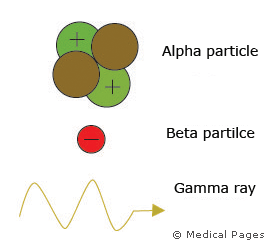
As the unstable nucleus changes, it gives off radiation and is said to be radioactive. This radiation can be given off as alpha particles, which include two protons and two neutrons (nucleus of helium atom), beta particles (electrons) and gamma rays (extremely high frequency electromagnetic radiation).
- Large atomic nuclei, with more than 83 protons are unstable. Uranium and plutonium are examples of such elements, however some smaller atomic nuclei may also be radioactive. Even hydrogen, has a radioactive isotope, known as tritium. Tritium contains one proton and two neutrons.

