Question #32c46
1 Answer
Oct 28, 2015
Just like we have a place to live, in the world of subatomic particles, electrons also have their own place. Where they go is dictated by the electron configuration, which describes the electron arrangement within an atom
As per the Quantum Mechanics Principles, the ground state electronic configuration of the Neon atom is
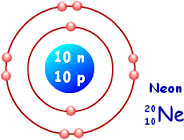
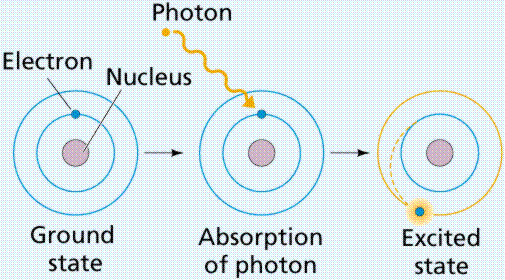
Neon atom has 2 electrons in First orbit and 8 electrons in the second orbit. If Electron picks up energy , electron can make a transition or jump from lower orbit to a higher orbit . This process is called excitation process and electron is said to be in excited state.