Question #6fda7
1 Answer
Yes, ammonia (
Let's examine ammonia's Lewis structure.
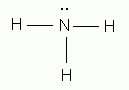
The possibility of hydrogen bonding is given by the presence of a lone pair of electrons on the nitrogen atom. Each lone pair present on the electronegative atom will attract one hydrogen atom from another ammonia molecule.
This happens because of the attraction that takes place between the partial negative charge that exists on the more electronegative atom and the partial positive charge that exists on the hydrogen atom.
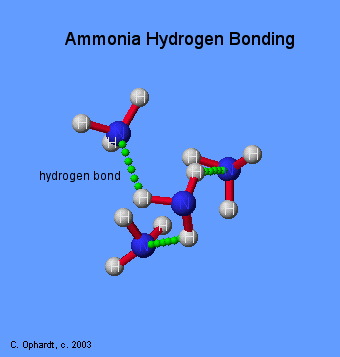
However, ammonia's capacity to form hydrogen bonds will be limited by the fact that the nitrogen atom has only one lone pair of electrons. As a result, in a group of ammonia molecules not all hydrogen atoms will form hydrogen bonds because of the limited supply of lone pairs
http://socratic.org/questions/can-any-one-explian-the-number-of-hydrogen-bonding-in-ammoina-molecule

