What is the net charge of a helium atom?
2 Answers
The net charge on the helium atom is zero.
Here's why that is. When it comes to net charge, the balance between electrons and protons will determine whether you have a negative charge - more electrons than protons, a positive charge - more protons than electrons, or no charge - equal numbers of electrons and protons.
The neutrons play no role in determining the charge of an atom because they're uncharged (neutral) particles.
In helium's case, you have 2 protons that are in the nucleus and 2 electrons that surround the nucleus.
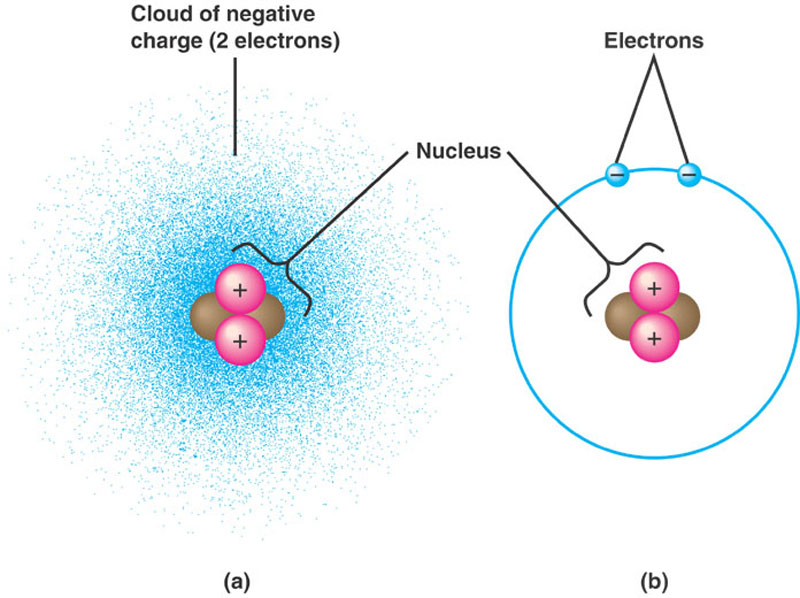
Each proton carries a +1 charge, while each electron carries a -1 charge. If the total positive charge and the total negative charge add up to give zero, the atom is said to be neutral. In helium's case, 2 protons and 2 electrons will give
Therefore, the helium atom is neutral.
Read more about how to determine net charge here:
http://socratic.org/questions/i-want-to-know-how-to-get-the-net-charge-for-each-element
A helium atom has no charge, which means it has an oxidation number of 0. Neutrons have no charge. Since the numbers of protons and electrons are equal, the positive charge of the protons and the negative charge of the electrons add up to zero.


